Human and Animal Experimentation Links
Urgent Action Needed Over Artificial Intelligence Risks to Human Rights

Urgent action is needed as it can take time to assess and address the serious risks this technology poses to human rights, warned the High Commissioner: "The higher the risk for human rights, the stricter the legal requirements for the use of AI technology should be". Ms. Bachelet also called for AI applications that cannot be used in compliance with international human rights law, to be banned. Read more...
US Lawmakers Concerned by Accuracy of Facial Recognition

US politicians expressed concerns about the accuracy and growing use of facial recognition software, at a hearing on Wednesday. The technology is being developed by firms including Amazon and Microsoft and increasingly used by law enforcement worldwide. Some facial recognition technologies misidentify women and people of colour. Civil liberties and privacy groups have raised concerns about how the data for these programs is being gathered. Read more...
The Ethics of Brain–Computer Interfaces
Link recommended by Linjiang Lou
"It becomes part of you,” Patient 6 said, describing the technology that enabled her, after 45 years of severe epilepsy, to halt her disabling seizures. Electrodes had been implanted on the surface of her brain that would send a signal to a hand-held device when they detected signs of impending epileptic activity. On hearing a warning from the device, Patient 6 knew to take a dose of medication to halt the coming seizure. Read more...
American Pragmatism as a Guide for Professional Ethical Conduct for Engineers
Link recommended by Christina Nnabuife
The ethical choices faced by engineers today are increasingly complex. Competing and conflicting ethical demands from clients, communities, employees, and personal objectives combine to suggest that engineers employ ethical approaches that are adaptive yet grounded in three concrete professional circumstances: first, that engineers apply unique professional skills in the service of a client, subject to protecting the public interest; second, that engineers advance the state of knowledge of their professional field through reflection, research, and sharing experience in journals and conferences, and third, that they develop new professionals by active mentoring. This paper examines five features of American pragmatism and suggests that its emphasis on specific, context-based ethical decision making can assist engineers in a postmodern setting. In particular, it considers the venues of interpersonal ethical choices, institutional ethical conflicts, and social choices that have ethical components. Pragmatism suggests that in such a complex ethical climate, there is a need for the co-evolution of judgment and action, for individual reflective judgment in particular situations, and for ceasing to search for a single, immutable principle for ethical choice. Read more...
Ethics Debate as Pig Brains Kept Alive Without a Body
Link recommended by Andrews Nartey
Researchers at Yale University have restored circulation to the brains of decapitated pigs, and kept the organs alive for several hours. Their aim is to develop a way of studying intact human brains in the lab for medical research. Although there is no evidence that the animals were aware, there is concern that some degree of consciousness might have remained. Details of the study were presented at a brain science ethics meeting held at the National Institutes of Health (NIH) in Bethesda in Maryland on 28 March. Read more...
Records from Controversial Twin Study Sealed at Yale Until 2065
Link recommended by Xiao Lin
In the depths of Yale’s library collections, records from a controversial study that separated twins and triplets at birth remain sealed, despite demands from the study’s participants to see their own files. The study, conducted by child psychiatrist Peter Neubauer throughout the 1960s and 70s, involved at least eight twins and a set of triplets who had been separated at birth at the now-defunct New York City adoption agency Louise Wise Services. Read more...
Rethinking Research Ethics: The Case of Postmarketing
Link recommended by Prince Kwaku Akowuah
Form the Nuremberg Code onward, the core mission of human subjects research ethics has been to protect study participants from infringements moti- vated by a zeal for medical progress. How- ever, with individuals, clinicians, and policy- makers increasingly dependent on scientific information for decision-making and with vast social resources invested in develop- ing and utilizing the fruits of research, actors have powerful incentives to coopt research for narrow ends. Contemplated revisions to human subjects research ethics policies in the United States (7) and existing policy in Canada (2) and the United Kingdom (5) fail to capture harms that, although they may not threaten participants, nonetheless undermine the social value of research. This is illus- trated by postmarketing (phase TV) research. As a corrective, research ethics should focus on safeguarding the integrity of research as a critical component of an evidence-driven, health information economy. Read more...
Scientists Have Put a Human Brain Gene into Monkeys. Have They Crossed the Line?
Link recommended by Linjiang Lou

A nightmare Planet of the Apes scenario, where intelligent apes displace humans, came to mind for many last week when a story broke out of China. Scientists genetically modified rhesus macaque monkeys with a human gene thought to play a role in the development of our big brains. And many in the west believe this research crossed an ethical red line. "The researchers were interested in what role genes played in human brain evolution," said Prof. Jim Tabery, a bioethicist and associate professor in the department of philosophy at the University of Utah in Salt Lake City, in conversation with Quirks & Quarks host Bob McDonald. Read more...
Ethics Creep: Governing Social Science Research in the Name of Ethics
Link recommended by Prince Kwaku Akowuah
This article presents an analysis of the Canadian ethics review process by a member of a Research Ethics Board. The author suggests that the new formal system for regulating the ethical conduct of scholarly research is experiencing a form of “ethics creep.” This is characterized by a dual process whereby the regulatory system is expanding outward to incorporate a host of new activities and institutions, while at the same time intensifying the regulation of activities deemed to fall within its ambit. These tendencies are demonstrated through an analysis of: 1) the scope of research ethics protocols, 2) the concept of “harm” employed by these boards, 3) the use of informed consent provisions, and 4) the presumption that research participants will remain anonymous. To accentuate the nature of this ethics creep, comparisons are made between the ways in which identical knowledge generation activities are governed within journalism and the academy. The conclusion suggests that one effect of the increasingly formalized research ethics structure is to rupture the relationship between following the rules and acting ethically. Some of the reasons for this “creep” are highlighted along with the risks that it poses for scholarship. Read more...
The Untold Story of the ‘Circle of Trust’ Behind the World’s First Gene-Edited Babies
Link recommended by Ashley Cruz
On 10 June 2017, a sunny and hot Saturday in Shenzhen, China, two couples came to the Southern University of Science and Technology (SUSTech) to discuss whether they would participate in a medical experiment that no researcher had ever dared to conduct. The Chinese couples, who were having fertility problems, gathered around a conference table to meet with He Jiankui, a SUSTech biophysicist. Then 33, He (pronounced "HEH") had a growing reputation in China as a scientist-entrepreneur but was little known outside the country. "We want to tell you some serious things that might be scary," said He, who was trim from years of playing soccer and wore a gray collared shirt, his cuffs casually unbuttoned. Read more...
Outrage Intensifies Over Claims Of Gene-Edited Babies
Link recommended by Syed Sajid Zaidi
Ever since a Chinese scientist rocked the world by claiming he had created gene-edited twin girls, international outrage has only intensified. "Everything that's emerged over the last week only adds to the concern about this having been a deeply unfortunate, misguided misadventure of the most dramatic sort," says Francis Collins, director of the U.S. National Institutes of Health. "It was shocking at the time. A week later, it's still shocking." Read more...
Needed: Closer Scrutiny of Clinical Trials
Link recommended by Bhavatharini Ramakrishnan
How many people know that eight patients in Hyderabad who were administered recombinant streptokinase to test its efficacy and safety have died? According to the Genetic Engineering Approval Committee (GEAC), the trial was being conducted by the drug’s manufacturer Shantha Biotechnics without taking clearance. Not surprisingly, the company denies the allegation claiming that it had taken permission from the Drugs Controller General, India (DCGI). In this game of passing the buck, no one is shedding any tears on the lives lost or compensating the families of those whose loved ones have died. Without any independent enquiry, the death of ‘trial subjects’, as they are impersonally called, has been attributed to ’causes other than the use’ of the drug! Read more...
The Science And Ethics Of Research On Chimps
Link recommended by Christina Nnabuife
The Southwest Foundation for Biomedical Research in San Antonio, Texas is one of only a handful of sites around the world that conduct medical research on great apes. Scientists say their research is conducted humanely, but many animal rights groups say testing on chimps is unneeded and unethical. Read more...
Consenting Adults? Not Necessarily...
Link recommended by Bhavatharini Ramakrishnan
In October 2003, China became the first country to move gene therapy into the medical mainstream, when it approved for general clinical use a treatment for head and neck squamous cancer. The feat was a symbol of the country's biomedical ambitions, and was lauded in the Chinese media. But just a few months later, a story emerged that revealed a grimmer aspect of China's rapid development in biomedicine. Read more...
E-cigarettes and the Paradox of Technology
Link recommended by Mariajose J Perez

Technology is a paradoxical thing. Human beings are tool-making animals, and have always looked to their abilities to craft things from the natural world to solve problems of living. Most of our knowledge of our early ancestors are reflected in the tools and technologies they have left behind. We use technologies to solve the problems of living, and so the things we create -arrowheads, cooking utensils, bone flutes, jewelry – can teach us about a culture’s priorities, needs, aesthetics, and resources. Read more...
Institutional Responsibility and the Flawed Genomic Biomarkers at Duke University: A Missed Opportunity for Transparency and Accountability
Link recommended by Augustine Nyarko
When there have been substantial failures by institutional leadership in their oversight responsibility to protect research integrity, the public should demand that these be recognized and addressed by the institution itself, or the funding bodies. This commentary discusses a case of research failures in developing genomic predictors for cancer risk assessment and treatment at a leading university. In its review of this case, the Office of Research Integrity, an agency within the US Department of Health and Human Services, focused their report entirely on one individual faculty member and made no comment on the institution’s responsibility and its failure to provide adequate oversight and investigation. These actions missed an important opportunity to emphasize the institution’s critical responsibilities in oversight of research integrity and the importance of institutional transparency and accountability. Read more...
Most Experimental Drugs are Tested Offshore–Raising Concerns about Data
Link recommended by Wei Hau Lew

The clinical trial for a herpes vaccine flouted just about every norm in the book: American patients were flown in to the Caribbean island of St. Kitts for experimental injections. Local authorities didn’t give permission. Nor did the Food and Drug Administration. Nor did a safety panel. That’s why the trial — run by a startup that has since received funding from billionaire investor Peter Thiel — prompted widespread alarm and censure when it was reported last week by Kaiser Health News. Read more...
Clinical Trials Have Far Too Little Racial and Ethnic Diversity
Link recommended by Wei Hau Lew

Nearly 40 percent of Americans belong to a racial or ethnic minority, but the patients who participate in clinical trials for new drugs skew heavily white—in some cases, 80 to 90 percent. Yet nonwhite patients will ultimately take the drugs that come out of clinical studies, and that leads to a real problem. The symptoms of conditions such as heart disease, cancer and diabetes, as well as the contributing factors, vary across lines of ethnicity, as they do between the sexes. If diverse groups aren’t part of these studies, we can’t be sure whether the treatment will work in all populations or what side effects might emerge in one group or another. Read more...
The Ethical Quandary of Human Infection Studies
Link recommended by Christine Pham
In February of last year, 64 healthy adult Kenyans checked into a university residence in the coastal town of Kilifi. After a battery of medical tests, they proceeded, one by one, into a room where a doctor injected them with live malaria parasites. Left untreated, the infection could have sickened or even killed them, since malaria claims hundreds of thousands of lives every year. Read more...
To Regulate or Not to Regulate? The Future of Animal Ethics in Experimental Research with Insects
Link recommended by Daniel Bao
Regulatory ethical frameworks governing animal experimentation are a hallmark of modern biology. While most countries have ethical standards regarding the use of animals for scientific purposes, experiments involving insects are not included in these standards. With studies in recent years suggesting that insects may possess faculties akin to emotive states, there is growing discussion surrounding the ethi-cal implications of scientific experimentation involving insects. This paper explores some of the current evidence for the ability of insects to experience emotive states and highlights how current formal animal experimentation ethics frameworks are unnecessary for governing the use of insects for scientific purposes. At its conclu-sion, this paper discusses ways in which scientists can, and should, uniformly max-imise the welfare of insects used in their experiments in a way that is of benefit to their science as well as to the dignity and welfare of their study organisms. Read more...
Europe’s biggest research fund cracks down on ‘ethics dumping’
Link recommended by Yumna Ali
Last year, President Bush cast the first veto of his presidency when Congress tried to ease the restriction on federal funding of embryonic stem cell research. Following the recent passage by both houses of Congress of the Stem Cell Research Enhancement Act of 2007, which would permit federal funding of research using donated surplus embryonic stem cells from fertility clinics, the president has once again threatened a veto. Because neither the House nor the Senate had sufficient votes to override a presidential veto, it appears unlikely this new bill will be enacted into law, further stalling the pace of this research. “This bill crosses a moral line that I and others find troubling,” stated Bush, following the Senate’s vote. Read more...
Examining the ethics of embryonic stem cell research
Link recommended by Angelie Ross
Last year, President Bush cast the first veto of his presidency when Congress tried to ease the restriction on federal funding of embryonic stem cell research. Following the recent passage by both houses of Congress of the Stem Cell Research Enhancement Act of 2007, which would permit federal funding of research using donated surplus embryonic stem cells from fertility clinics, the president has once again threatened a veto. Because neither the House nor the Senate had sufficient votes to override a presidential veto, it appears unlikely this new bill will be enacted into law, further stalling the pace of this research. “This bill crosses a moral line that I and others find troubling,” stated Bush, following the Senate’s vote. Read more...
Experimenting on animals
Link recommended by Sunny Patil
Animal experiments are widely used to develop new medicines and to test the safety of other products. Many of these experiments cause pain to the animals involved or reduce their quality of life in other ways. If it is morally wrong to cause animals to suffer then experimenting on animals produces serious moral problems. Read more...
Did a study of 3D bioprint me: a socioethical view of bioprinting human organs and tissues
Link recommended by Tanooha Veeramachaneni
In this article, we review the extant social science and ethical literature on three-dimensional (3D) bioprinting. 3D bioprinting has the potential to be a ‘game-changer’, printing human organs on demand, no longer necessitating the need for living or deceased human donation or animal transplantation. Although the technology is not yet at the level required to bioprint an entire organ, 3D bioprinting may have a variety of other mid-term and short-term benefits that also have positive ethical consequences, for example, creating alternatives to animal testing, filling a therapeutic need for minors and avoiding species boundary crossing. Despite a lack of current socioethical engagement with the consequences of the technology, we outline what we see as some preliminary practical, ethical and regulatory issues that need tackling. These relate to managing public expectations and the continuing reliance on technoscientific solutions to diseases that affect high-income countries. Avoiding prescribing a course of action for the way forward in terms of research agendas, we do briefly outline one possible ethical framework ‘Responsible Research Innovation’ as an oversight model should 3D bioprinting promises are ever realised. 3D bioprinting has a lot to offer in the course of time should it move beyond a conceptual therapy, but is an area that requires ethical oversight and regulation and debate, in the here and now. The purpose of this article is to begin that discussion. Read more...
Did a study of Indonesian people who spend most of their days under water violate ethical rules?
Link recommended by Yumna Ali

In April, a paper showing why Indonesia's Bajau people are such great divers drew worldwide attention as a striking example of recent human evolution. But the study, published in Cell, has created a different kind of stir in Indonesia, where some say it is an example of "helicopter research" carried out by scientists from rich countries with little consideration for local regulations and needs. Read more...
Will CRISPR Germline Engineering Close the Door to an Open Future?
Link recommended by Cole Hudson
Being inherently different from any other lifesaving organ transplant, uterine transplantation does not aim at saving lives but supporting the possibility to generate life. Unlike the kidneys or the liver, the uterus is not specifically a vital organ. Given the non-lifesaving nature of this procedure, questions have been raised about its feasibility. The ethical dilemma revolves around whether it is worth placing two lives at risk related to surgery and immunosuppression, amongst others, to enable a woman with absolute uterine factor infertility to experience the presence of an organ enabling childbirth. In the year 2000, the first uterine transplantation, albeit unsuccessful, was performed in Saudi Arabia from where it has spread to the rest of the world including Sweden, the United States and now recently India. The procedure is, however, still in the preclinical stages and several ethical, legal, social and religious concerns are yet to be addressed before it can be integrated into the clinical setting as standard of care for women with absolute uterine factor infertility. Read more...
Uterine Transplant: A Risk to Life or a Chance for Life?
Link recommended by Jade Hostetter
Being inherently different from any other lifesaving organ transplant, uterine transplantation does not aim at saving lives but supporting the possibility to generate life. Unlike the kidneys or the liver, the uterus is not specifically a vital organ. Given the non-lifesaving nature of this procedure, questions have been raised about its feasibility. The ethical dilemma revolves around whether it is worth placing two lives at risk related to surgery and immunosuppression, amongst others, to enable a woman with absolute uterine factor infertility to experience the presence of an organ enabling childbirth. In the year 2000, the first uterine transplantation, albeit unsuccessful, was performed in Saudi Arabia from where it has spread to the rest of the world including Sweden, the United States and now recently India. The procedure is, however, still in the preclinical stages and several ethical, legal, social and religious concerns are yet to be addressed before it can be integrated into the clinical setting as standard of care for women with absolute uterine factor infertility. Read more...
The ethics of animal research
Link recommended by Sunny Patil
Animal research has had a vital role in many scientific and medical advances of the past century and continues to aid our understanding of vari- ous diseases. Throughout the world, peo- ple enjoy a better quality of life because of these advances, and the subsequent development of new medicines and treat- ments—all made possible by animal research. However, the use of animals in scientific and medical research has been a subject of heated debate for many years in the UK. Opponents to any kind of ani- mal research—including both animal- rights extremists and anti-vivisectionist groups—believe that animal experimenta- tion is cruel and unnecessary, regardless of its purpose or benefit. There is no mid- dle ground for these groups; they want the immediate and total abolition of all animal research. If they succeed, it would have enormous and severe consequences for scientific research. Read more...
Emerging stem cell ethics
Link recommended by Yumna Ali
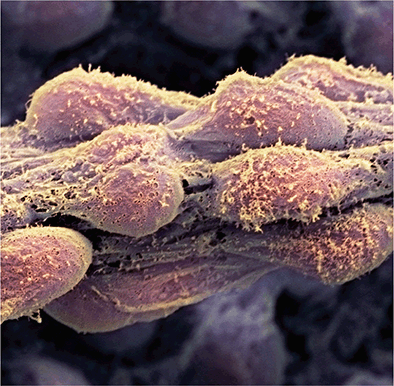
It has been 20 years since the first derivation of human embryonic stem cells. That milestone marked the start of a scientific and public fascination with stem cells, not just for their biological properties but also for their potentially transformative medical uses. The next two decades of stem cell research animated an array of bioethical debates, from the destruction of embryos to derive stem cells to the creation of human-animal hybrids. Ethical tensions related to stem cell clinical translation and regulatory policy are now center stage and a topic of global discussion this week at the International Society for Stem Cell Research (ISSCR) annual meeting in Melbourne, Australia. Care must be taken to ensure that entry of stem cell–based products into the medical marketplace does not come at too high a human or monetary price. Read more...
Mental capacity to consent to research? Experiences of consenting adults with intellectual disabilities and/or autism to research
Link recommended by Madeleine Lu
Adults with intellectual disabilities and/or autism are often excluded from participating in health and healthcare research. Understanding study information, which is an important aspect of demonstrating capacity to give informed consent, can be a particular challenge. This study surveyed clinical researchers to discover: (i) their experiences of assessing mental capacity for research; (ii) what methods they used to facilitate the inclusion of adults with intellectual disabilities and/or autism; and (iii) their views about a proposal to develop new resources to facilitate mental capacity judgements with adults with intellectual disabilities and/or autism for informed consent for research.
Clinical researchers in North East England who conduct research with NHS patients with intellectual disabilities and/or autism were invited to participate in a 22-item self-completed semi-structured questionnaire survey, either online or on paper. Results: Twenty-one clinicians completed the survey (response rate 30.4%). Participants reported on 18 research studies which included people with intellectual disabilities and/or autism. In many studies people who lacked capacity to give informed consent were excluded, and often shortcuts were taken in judging capacity. Limited adaptations to support capacity were used. Respondents welcomed the proposal of developing assistive resources that could support capacity judgements and informed consent to research. Read more...
Help, hope, and hype: Ethical dimensions of neuroprosthetics
Link recommended by Eugene A Osae

Brain-controlled prosthetic robots that restore independent activities of daily living to paralyzed people are about to enter everyday life environments (1). The regained ability to grasp a cup of coffee, hand over a credit card, or sign a document with a pen (1) enhances the independence and self-determination of severely paralyzed individuals. However, introducing devices controlled via brain-machine interfaces (BMIs) into everyday environments, possibly enhancing the capabilities of able-bodied people to interact with digital devices, raises a number of ethical and social challenges in the areas of (i) autonomy, responsibility, and accountability; (ii) data security and privacy; and (iii) managing end-user expectations about a promising field of medical advances. We here take a closer look at these issues and suggest some possible answers to addressing them. Read more...
The ethical neuroscientist
Link recommended by Swetcha Chowdary Karnati
The increase in research carried out by private companies raises concerns about ethical standards for human subjects. A recent paper1 and an accompanying commentary2 illustrate how seriously the academic community takes the ethical decisions involved in research on human subjects. However, neuroscience research is increasingly being done in the private sector, which is not obligated to follow the same guidelines as academic institutions. To avoid concerns similar to those raised about private-sector medical research, commercial neuroscience should follow standard ethical guidelines. The new study1 compared abandoned children reared in Romanian orphanages to similar children who were moved to foster care. Unsurprisingly, cognitive development in children who remained institutionalized was markedly lower than that of never-institutionalized children or of children who were removed from institutions and placed into foster care. The authors conclude that early institutionalization has serious negative consequences on child development and that family placement should be preferred. Read more...
A cross-sectional study: Need of equal respect for all professionals in the Institutional Ethics Committees' composition
Link recommended by Moawiz M Saeed
The composition of the Institutional Ethics Committee (IEC) with an equal respect plays a major role in evaluating research proposals to ensure the safety of the subjects and ethical quality of research project. It is mandatory that all research projects with an involvement of human subjects should be approved by the IEC before commencement. Methods include a cross-sectional study, the general information and member composition, multidisciplinary nature, independent performance, competence, decision capability, professionally biased of IECs in health research institutions of Punjab, India. A structured self-administered questionnaire was developed and provided to all health/research institutions providing the graduate/postgraduate education in medical sciences under the jurisdiction of Baba Farid University of Health Sciences, Faridkot, Punjab, India. Read more...
Genome Editing
Link recommended by Akshata Divakar Goudar
Most of the ethical discussions related to genome editing center around human germline editing. This is because changes made in the germline would be passed down to future generations. The debate about genome editing is not a new one but has regained attention following the discovery that CRISPR has the potential to make such editing more accurate and even "easy" in comparison to older technologies. Bioethicists and researchers generally believe that human genome editing for reproductive purposes should not be attempted at this time, but that studies that would make gene therapy safe and effective should continue.1,2 Most stakeholders agree that it is important to have continuing public deliberation and debate to allow the public to decide whether or not germline editing should be permissible. As of 2014, there were about 40 countries that discouraged or banned research on germline editing, including 15 nations in Western Europe, because of ethical and safety concerns.3 There is also an international effort led by the US, UK, and China to harmonize regulation of the application of genome editing technologies. This effort officially launched in December 2015 with the International Summit on Human Gene Editing in Washington, DC. Read more...
Researchers grapple with the ethics of testing brain implants
Link recommended by Moawiz M Saeed and Wenpei Tang

In 2003, neurologist Helen Mayberg of Emory University in Atlanta began to test a bold, experimental treatment for people with severe depression, which involved implanting metal electrodes deep in the brain in a region called area 25. The initial data were promising; eventually, they convinced a device company, St. Jude Medical in Saint Paul, to sponsor a 200-person clinical trial dubbed BROADEN. This month, however, Lancet Psychiatry reported the first published data on the trial’s failure. The study stopped recruiting participants in 2012, after a 6-month study in 90 people failed to show statistically significant improvements between those receiving active stimulation and a control group, in which the device was implanted but switched off. Read more...
Culture, law, ethics, and social implications: Is society ready for advanced genomic medicine?
Link recommended by Angie Stephanie De La Cruz
In recent years, rapid and continuing advances in our knowledge and interpretation of information on the human genome have transformed our understanding of human health, medical diagnostics, and therapy. Accessing information on genetic material is now quicker, cheaper, and more widespread than at any other time in our history. As innovations and discoveries involving human genetic material continue, there will be an ongoing necessity to carefully integrate advanced genomic medicine (AGM), including personalised genetic diagnostics and intervention, into the wider community. AGM offers the potential of personalised genomic profiling that enables the diagnosis of disease and targeted treatment, yet despite the advantages of such technology, there are issues with accurate diagnosis and interpretation of data. Even experts struggle with the complex nature of the data that is currently gathered. Read more...
Four ethical priorities for neurotechnologies and AI
Link recommended by Angie Stephanie De La Cruz and Khob Raj Bhandari

Consider the following scenario. A paralysed man participates in a clinical trial of a brain–computer interface (BCI). A computer connected to a chip in his brain is trained to interpret the neural activity resulting from his mental rehearsals of an action. The computer generates commands that move a robotic arm. One day, the man feels frustrated with the experimental team. Later, his robotic hand crushes a cup after taking it from one of the research assistants, and hurts the assistant. Apologizing for what he says must have been a malfunction of the device, he wonders whether his frustration with the team played a part. This scenario is hypothetical. But it illustrates some of the challenges that society might be heading towards. Read more...
Is It Ethical to Edit an Embryo's Genes?
Link recommended by Wenpei Tang
Warning the world about the potential perils of genetic engineering, physician-novelist Robin Cook brought us his medical thriller Mutation in 1990. At the time, his foreboding tale about the perfect designer baby–turned–evil genius seemed like pure science fiction. A quarter century later, though, gene-editing tools are being perfected in labs throughout the world, and some fear that Cook's fantasy could someday be reality. The first step in that scenario, human germline editing, is already a reality. Germline editing refers to genetically engineering a genome in ways that are heritable—as in, editing reproductive cells or embryos. And while gene therapy for ill, consenting adults is both promising and widely accepted as safe and ethical, in part because it involves nonheritable changes in somatic cell lines, editing the genes of embryos to create genetically modified human beings has raised significant concerns. Read more...
Editing human embryos with CRISPR is moving ahead – now’s the time to work out the ethics
Link recommended by Khob Raj Bhandari
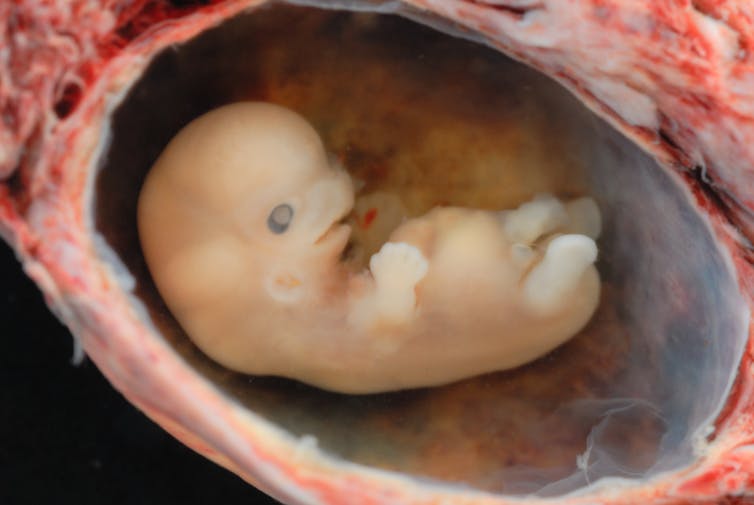
The announcement by researchers in Portland, Oregon that they’ve successfully modified the genetic material of a human embryo took some people by surprise. With headlines referring to “groundbreaking” research and “designer babies,” you might wonder what the scientists actually accomplished. This was a big step forward, but hardly unexpected. As this kind of work proceeds, it continues to raise questions about ethical issues and how we should we react. Read more...
Another Study Of Preemies Blasted Over Ethical Concerns
Link recommended by Snigdha Reddy Dodla

For the second time in four months, the consumer group Public Citizen is alleging that a large, federally funded study of premature infants is ethically flawed. Both complaints raise a big issue that's certain to get more attention beyond these particular studies: What's the ethically right way to do research on the validity of the usual care that doctors provide every day. Read more...
Self-Driving Cars and Engineering Ethics: The Need for a System Level Analysis
Link recommended by Dalia L Lezzar

The literature on self-driving cars and ethics continues to grow. Yet much of it focuses on ethical complexities emerging from an individual vehicle. That is an important but insufficient step towards determining how the technology will impact human lives and society more generally. What must complement ongoing discussions is a broader, system level of analysis that engages with the interactions and effects that these cars will have on one another and on the socio-technical systems in which they are embedded. To bring the conversation of self-driving cars to the system level, we make use of two traffic scenarios which highlight some of the complexities that designers, policymakers, and others should consider related to the technology. We then describe three approaches that could be used to address such complexities and their associated shortcomings. We conclude by bringing attention to the “Moral Responsibility for Computing Artifacts: The Rules”, a framework that can provide insight into how to approach ethical issues related to self-driving cars. Read more...
THE GREAT SILVER SPRING MONKEY DEBATE
Link recommended by Snigdha Reddy Dodla
ON SEPTEMBER 11, 1981, in an unprecedented raid on a scientific laboratory, Montgomery County police seized 17 monkeys being used for neurological research. Nearly a decade later, the battle over animal rights rages on - and the surviving monkeys are caught in the crossfire. THE FIRST THING THAT HIT THEM WAS THE SMELL. As soon as the Montgomery County police stepped through the door of the Institute for Behavioral Research in Silver Spring, they caught a whiff of an unmistakable ammonia odor that jolted their nostrils like smelling salts. Out in the office area, it merely smelled "like a pet shop," as one cop recalls, but as they got closer to the colony room, where the monkeys were kept, it got stronger and stronger. Read more...
Ethics and Artificial Intelligence: The Moral Compass of a Machine
Link recommended by Sujata Rijal
:format(webp)/cdn.vox-cdn.com/uploads/chorus_image/image/49557197/robot-ethics_john-williams-rus.0.jpg)
The question of robotic ethics is making everyone tense. We worry about the machine’s lack of empathy, how calculating machines are going to know how to do the right thing, and even how we are going to judge and punish beings of steel and silicon. Personally, I do not have such worries. I am less concerned about robots doing wrong, and far more concerned about the moment they look at us and are appalled at how often we fail to do right. I am convinced that they will not only be smarter than we are, but have truer moral compasses, as well. Read more...
Use of E-Cigarettes by Young People Is Major Concern, Surgeon General Declares
Soaring use of e-cigarettes among young people “is now a major public health concern,” according to a report published Thursday from the United States Surgeon General. It is the first comprehensive look on the subject from the nation’s highest public-health authority, and it finds that e-cigarettes are now the most commonly used tobacco product among youths, surpassing tobacco cigarettes. E-cigarettes, which turn nicotine into inhalable vapor, can harm developing brains of teenagers who use them, and also can create harmful aerosol for people around the user, the report said, citing studies in animals. Read more...
Letting a baby play on an iPad might lead to speech delays, study says
Anyone raising a child today has likely fretted about screen time and wondered about the impact of devices on our kids. Does the technology affect their brains? Does it limit their social development? Could it harm them emotionally? Could it delay when they start talking? I had never thought about that last question until a new study, released Thursday and being presented at the 2017 Pediatric Academic Societies Meeting, revealed some striking findings. The study found that the more time children between the ages of six months and two years spent using handheld screens such as smartphones, tablets and electronic games, the more likely they were to experience speech delays. Read more...
Instagram worst social media app for young people's mental health

Instagram is the most detrimental social networking app for young people's mental health, followed closely by Snapchat, according to a new report by the Royal Society for Public Health in the UK. Their study, #StatusofMind, surveyed almost 1,500 young people aged 14 to 24 on how certain social media platforms impact health and well-being issues such as anxiety, depression, self-identity and body image. YouTube was found to have the most positive impact, while Instagram, Snapchat, Facebook and Twitter all demonstrated negative affects overall on young people's mental health. Read more...
Your smartphone may be hurting your sleep

You love your smartphone, but it may be ruining your ZZZ's. Use of these devices, especially near bedtime, is associated with worse quality of sleep, according to a new study. "When we looked at smartphone use around the time when participants reported they went to bed, more smartphone use around that time in particular was associated with a longer time to fall asleep and worse sleep quality during the night," said Dr. Gregory Marcus, author of the study and an associate professor of medicine at the University of California, San Francisco. Read more...
3+ hours daily screen time linked to diabetes risk factors for kids
Daily screen time of three or more hours is linked to several risk factors associated with the development of diabetes in children, finds research published online in the Archives of Disease in Childhood. These include adiposity, which describes total body fat, and, crucially, insulin resistance, which occurs when cells fail to respond to insulin, the hormone produced by the pancreas to control levels of blood glucose. Previous research suggests that spending a lot of time glued to a screen is linked to a heightened risk of weight gain and type 2 diabetes among adults. But it is not clear if children might also be at risk, particularly as recent trends indicate that the amount of time they spend watching TV and using computers, games consoles, tablets and smartphones is on the rise. The researchers therefore assessed a sample of nearly 4500 9-10 year old pupils from 200 primary schools in London, Birmingham, and Leicester for a series of metabolic and cardiovascular risk factors. Read more...
Experiment to raise the dead blocked in India
Link recommended by Felix Yemanyi
The Indian Council of Medical Research (ICMR) has derailed a controversial experiment that would seek to revive brain-dead accident victims. On 11 November, ICMR’s National Institute of Medical Statistics removed the “ReAnima” trial from India’s clinical trial registry. In May, Himanshu Bansal, an orthopedic surgeon at Anupam Hospital in the north Indian state of Uttarakhand, announced plans to give around 20 brain-dead people a mix of interventions including injections of mesenchymal stem cells and peptides, and transcranial laser stimulation and median nerve stimulation. Transcranial laser stimulation involves shining pulses of near-infrared light into the brain; median nerve stimulation is the electrical stimulation of a major nerve that runs from the neck to the arm. Both techniques have been shown to improve cognition in patients with traumatic brain injury. Bioquark, a biotech firm based in Philadelphia, Pennsylvania, had agreed to supply the trial with peptides that are said to help regenerate brain cells. Read more...
Second Thoughts of an Animal Researcher
Link recommended by Audrey Lees
Five years ago, the National Institutes of Health all but ended biomedical and behavioral research on chimpanzees, concluding that, as the closest human relative, they deserved “special consideration and respect.” But chimpanzees were far from the only nonhuman primates used in research then, or now. About 70,000 other primates are still living their lives as research subjects in labs across the United States. On Wednesday, the N.I.H. will hold a workshop on “continued responsible research” with these animals. This sounds like a positive development. But as someone who spent decades working almost daily with macaque monkeys in primate research laboratories, I know firsthand that “responsible” research is not enough. What we really need to examine is the very moral ground of animal research itself. Read more...
Ethical Issues and Vaccines
Link recommended by Sudan Puri
Vaccines are responsible for many global public health successes, such as the eradication of smallpox and significant reductions in other serious infections like polio and measles. Even so, vaccinations have also long been the subject of various ethical controversies. The key ethical debates related to vaccine regulation, development, and use generally revolve around (1) mandates, (2) research and testing, (3) informed consent, and (4) access disparities. Read more...
An N.Y.U. Study Gone Wrong, and a Top Researcher Dismissed
Link recommended by Felix Yemanyi
New York University’s medical school has quietly shut down eight studies at its prominent psychiatric research center and parted ways with a top researcher after discovering a series of violations in a study of an experimental, mind-altering drug. A subsequent federal investigation found lax oversight of study participants, most of whom had serious mental issues. The Food and Drug Administration investigators also found that records had been falsified and researchers had failed to keep accurate case histories. Read more...
HEALTH-INDIA: Prime Destination for Unethical Clinical Trials
Link recommended by Indu Sankaranayanan
Lack of regulation, accountability, low costs of operation and wide availability of target participants are reasons why multinational drug companies, researchers and institutions are increasingly basing their clinical trials in India. An estimated 40 percent of all clinical trials now take place in Asia, Eastern Europe, central and south America. “There is no compulsory registration system for clinical trials in these countries and many do not follow European directives in their operations”, says Dr. Jacob Sijtsma of the Netherlands-based WEMOS, an advocacy health organisation tracking clinical trials in developing countries. Read more...
Unnecessary Roughness: PETA sacks NFL’s Animal Testing
Link recommended by George Panagopoulos
PETA has posted an action alert against the NFL’s alleged funding of animal testing. The organization is petitioning to end the practice of simulating sports injuries on mice, rats, and even dogs. Their website displays a shockingly graphic image of a rat being hit in the head with a metal rod as part of the alleged experiments. According to PETA, the research is being conducted at several universities as well as private labs. They suggest the NFL is funding the research due to recent legal matters concerning injured football players. PETA asserts the testing is not only unethical but also inefficient. Read more...
The Best-Selling, Billion-Dollar Pills Tested on Homeless People
Link recommended by Sonia Shirwadkar
Two years ago, on a gray January afternoon, I visited the Ridge Avenue homeless shelter in Philadelphia. I was looking for poor people who had been paid to test experimental drugs. The streets outside the shelter were lined with ruined buildings and razor wire, and a pit bull barked behind a chain-link fence. A young guy was slumped on the curb, glassy-eyed and shaky. My guide, a local mental health activist named Connie Schuster, asked the guy if he was okay, but he didn’t answer. “My guess is heroin,” she said. Read more...
10 Bizarre Psychology Experiments that Completely Crossed the Line
Link recommended by Samuel Adade
Psychological experiments can be key to understanding what makes people tick, yet some individuals have gone about their research in rather unusual – and sometimes morally dubious – ways. Furthermore, while these researchers’ findings may increase the sum of knowledge on human behavior, the methods that a number of psychologists have used in order to test theories have at times overstepped ethical boundaries or might even appear somewhat sadistic. Those taking part in such studies have not always escaped unscathed, either; in fact, as a result some have suffered lasting emotional damage – or worse. Read more...
Social transfer of pain in mice
A complex relationship exists between the psychosocial environment and the perception and experience of pain, and the mechanisms of the social communication of pain have yet to be elucidated. The present study examined the social communication of pain and demonstrates that “bystander” mice housed and tested in the same room as mice subjected to inflammatory pain or withdrawal from morphine or alcohol develop corresponding hyperalgesia. Olfactory cues mediate the transfer of hyperalgesia to the bystander mice, which can be measured using mechanical, thermal, and chemical tests. Hyperalgesia in bystanders does not co-occur with anxiety or changes in corticosterone and cannot be explained by visually dependent emotional contagion or stress-induced hyperalgesia. These experiments reveal the multifaceted relationship between the social environment and pain behavior and support the use of mice as a model system for investigating these factors. In addition, these experiments highlight the need for proper consideration of how experimental animals are housed and tested. Read more...
How are all those screens changing kids' behavior?

In a recent op-ed in the New York Times, businessman and author Tony Schwartz offered an honest yet troubling account of what he calls his "addiction" to technology.
The medical and research fields have not yet come to a clear consensus on what constitutes technology "addiction" and which factors distinguish a true technology addiction disorder from problematic use or just bad habits. But the behavior Schwartz describes is remarkably familiar to those of us who are on our devices more than we know we ought to be. Read more...
China Is Building a Giant Animal-Cloning Factory to Feed the Masses
Link recommended by Patrick Mullarkey
How do you feel about eating cloned meat?
While the rest of the world sorts out its feelings about the safety and ethics of cloning animals for food production, China is charging ahead and building the world’s largest animal cloning factory, set to begin operations in 2016. The 200 million yuan (over $31 million) commercial animal cloning center will be located in the Tianjin Economic-Technological Development Area, a government-sponsored business area about 100 miles from Beijing.
Feelings are decidedly mixed about whether we should be eating cloned meat. On one hand, the European Parliament recently voted by a large margin to outlaw the sale of cloned livestock. On the other hand, the FDA has said that “there are no complications that are unique to cloning” and the meat is safe to eat, despite evidence that cloned animals may not live as long as those birthed in the old-fashioned way. In the US, no special labeling is required for cloned meat, and no one really seems to know how common the practice of cloning meat for food is. Still, some say that most of the cattle cloned in the US are used to breed stock and are not sold for food. Read more...
Hopkins faces $1B lawsuit over role in government study that gave subjects STDs
Link recommended by Laura Pardon
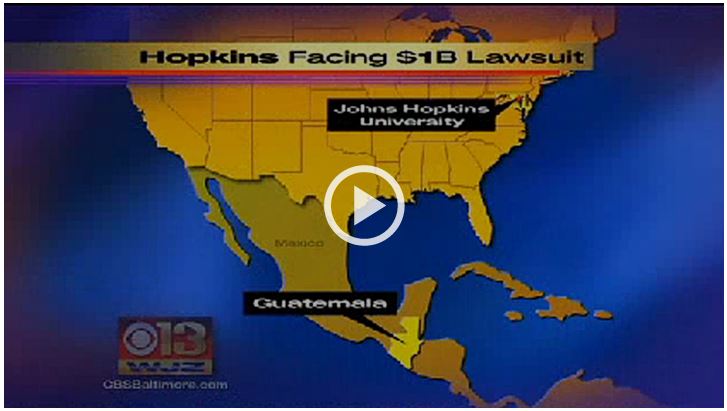
Narly 800 former research subjects and their families filed a billion-dollar lawsuit Wednesday against the Johns Hopkins University, blaming the institution for its role in 1940s government experiments in Guatemala that infected hundreds with syphilis, gonorrhea and other sexually transmitted diseases. Read more...
The Many Ethical Implications of Emerging Technologies
Link recommended by Laura Pardon

In the past four decades technology has fundamentally altered our lives: from the way we work to how we communicate to how we fight wars. These technologies have not been without controversy, and many have sparked intense debates that are often polarized or embroiled in scientific ambiguities or dishonest demagoguery.
The debate on stem cells and embryo research, for example, has become a hot-button political issue involving scientists, policy makers, politicians and religious groups. Similarly, the discussions on genetically modified organisms (GMOs) have mobilized civil society, scientists and policy makers in a wide debate on ethics and safety. The developments in genome-editing technologies are just one example that bioresearch and its impact on market goods are strongly dependent on social acceptance and cannot escape public debates of regulation and ethics. Moreover, requests for transparency are increasingly central to these debates, as shown by movements like Right to Know, which has repeatedly demanded the labeling of GMOs on food products. Read more...
'A robot is my friend': Can machines care for elderly?

With the world's elderly population growing rapidly, scientists are suggesting that robots could take on some of the burden of providing care, support and - most surprisingly - companionship.
A boy born today in Britain is expected to live on average to the age of 91, a girl to 94.
Worldwide 1.5 billion people over the age of 65 are expected to be around in 2050. Read more...
Human medical experiments go terribly wrong in "nightmare" TGN1412 drug trial
Link recommended by Disha Jain
"We told ya so" just doesn't cut it. Barely ten days after Truth Publishing warned the public about the dangers of medical experimentation on humans, a disastrous pharmaceutical experiment has left six men fighting for their lives.
The scene was a living medical hell, say witnesses. After being injected with the anti-inflammatory drug TGN1412, patients began tearing their shirts off, screaming that their heads were going to explode. One patient's head swelled to triple its normal size, and patients were soon passing out, vomiting, or screaming in sheer terror. Read more...
Testing Drugs on the Developing World
Link recommended by Rachel Funk

The purpose of clinical trials is to find out if the newest wonder drug is all that wonderful, and what kind of side effects we humans might expect. It worked on animals, but will this drug kill people, and/or turn them green? Every warning you see on a label is there because a test subject -- or 50 of them, or 500 of them -- have suffered that side effect. Read more...
Were Tuskegee & Willowbrook 'Studies in Nature'?
By David J. RothmanLink recommended by Tyler Watkins

The book jacket of Bad Blood, James Jones's recent account of the Tuskegee syphilis experiment, describes the project as one in which "science went mad" (New York: Free Press, 1981). Apparently the case is exceptional, an abberation from normal biomedical research behavior. But put the Tuskegee experiment alongside the Willowbrook experiments of the 1950s and 1960s, in which retarded and institutionalized children were injected with live hepatitus viruses, and clearly something other than "mad science" was at stake. Read More...
Study of Babies Did Not Disclose Risks, U.S. Finds
Link recommended by Emily Laird
A federal agency has found that a number of prestigious universities failed to tell more than a thousand families in a government-financed study of oxygen levels for extremely premature babies that the risks could include increased chances of blindness or death.
None of the families have yet been notified of the findings from the Office for Human Research Protections, which safeguards people who participate in government-financed research. But the agency’s conclusions were listed in great detail in a letter last month to the University of Alabama at Birmingham, the lead site in the study. In all, 23 academic institutions took part, including Stanford, Duke and Yale. Read More...
Unethical Clinical Trials Still Being Conducted in Developing Countries
By Michael CaromeLink recommended by Timothy Liu

In 1997, Public Citizen's Health Research Group brought widespread international attention to unethical clinical trials. The trials were testing new methods for preventing the spread of HIV infection from pregnant women to their babies before or after giving birth in developing countries in Africa, Asia and the Caribbean. In each of these trials, most of which were funded by the U.S. government, some women were randomly assigned to receive placebos or other treatments known to be ineffective, rather than a drug proven effective in preventing the spread of HIV infection from mother to baby.
Unfortunately, nearly 20 years later, similarly unethical trials continue to be conducted. One recent such trial in India, reported in The Lancet in June, evaluated an experimental vaccine for preventing a very common, potentially life-threatening viral infection called rotavirus. Two rotavirus vaccines have been available for the past decade: RotaTeq, marketed by Merck & Co., and Rotarix, marketed by GlaxoSmithKline. Large clinical trials of both vaccines demonstrated that they were highly effective in preventing rotavirus-induced gastroenteritis -- including the most severe cases -- and the need for hospitalization. Despite the availability of these vaccines, more than 2,000 children in the India trial received placebo injections of salt water rather than one of the available effective vaccines. Read More...
Deaths of three beagles spur ads decrying animal research in Denver
By Kieran NicholsonLink recommended by Sarah Jarudi

A national physicians group wanting an end to the use of animals as research subjects in scientific and medical studies has launched its latest advertising campaign in Denver, and it's centered on the deaths of three research dogs in Colorado.
The Physicians Committee for Responsible Medicine, a nonprofit organization formed in 1985 and based in Washington, D.C., has targeted Denver with ads about three beagles that died in 2011 at the CARE Research facility in Fort Collins. Read More...
Deception at Duke: Fraud in cancer care?
Link recommended by Sudad Kazzaz

Chemotherapy can be a tough road for people with cancer, often debilitating and even dangerous. Which is why five years ago, when Duke University announced that it had an advanced, experimental treatment that would match chemotherapy to a patient's own genetic makeup, it was hailed as the holy grail of cancer care. The scientist behind the discovery was Dr. Anil Potti, and soon Dr. Potti became the face of the future of cancer treatment at Duke, offering patients a better chance even with advanced disease. However, when other scientists set out to verify the results, they found many problems and errors. What our 60 Minutes investigation reveals is that Duke's so-called breakthrough treatment wasn't just a failure -- it may end up being one of the biggest medical research frauds ever. Watch the interview
How A Tilt Toward Safety Stopped A Scientist's Virus Research
Link recommended by Dinesh Majeti
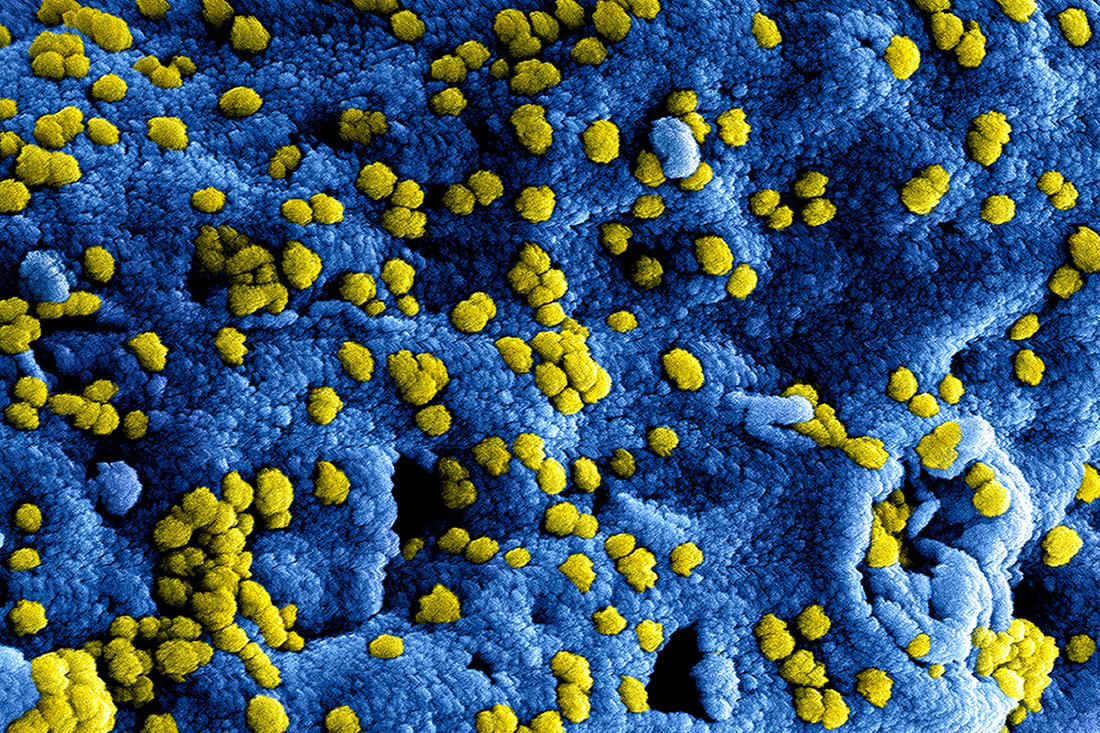
As cases of a worrisome respiratory virus continue to pop up in the Middle East, scientists who study it in the U.S. are struggling to understand how they'll be affected by a government moratorium on certain kinds of experiments.
One of those researchers is Ralph Baric, a virologist at the University of North Carolina School of Medicine in Chapel Hill. "Any virus that has pandemic potential, and that's any respiratory virus that emerges from animals, is a major public health concern," Baric says. Listen to the story
Are Placebos Ethical?
By John C. GoodmanLink recommended by Karl Kyeongan Kwon


The power of placebos to heal has long been recognized. Thomas Jefferson wrote about it. So did Benjamin Franklin. Debate over the ethics of placebos also has a long history – roughly 170 years. But there has been no resolution. Doctors are still in disagreement about whether it is ethical to proscribe a placebo without the patient’s consent. Of course if the patient knows the therapy is a placebo, much of its effectiveness is lost. But there is also disagreement about the meaning of the word “consent.” Read more ...
Scientists condemn 'crazy, dangerous' creation of deadly airborne flu virus
Link recommended by Kwami Antwi-Boasiako
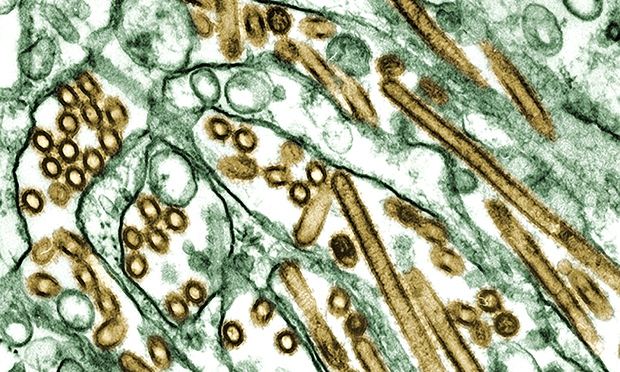
Scientists have created a life-threatening virus that closely resembles the 1918 Spanish flu strain that killed an estimated 50m people in an experiment labelled as "crazy" by opponents.
US researchers said the experiments were crucial for understanding the public health risk posed by viruses currently circulating in wild birds, but critics condemned the studies as dangerous and called on funders to stop the work. Read more ...
Internet outraged by Facebook's 'creepy' mood experiment
By Charles RileyLink recommended by Malcolm Dcosta


Everyone has a bad day on occasion. But what if Facebook made it worse -- on purpose, and without telling you?
Internet users have reacted angrily to news that Facebook researchers manipulated the content some users were shown in an attempt to gauge their emotional response.
For one week in early 2012, Facebook (FB, Tech30) changed the content mix in the News Feeds of almost 690,000 users. Some people were shown a higher number of positive posts, while others were shown more negative posts. Read more ...
Genetic engineering of animals: Ethical issues, including welfare concerns
By Elisabeth H. Ormandy, Julie Dale, Gilly GriffinLink recommended by Ashik Khatri

The genetic engineering of animals has increased significantly in recent years, and the use of this technology brings with it ethical issues, some of which relate to animal welfare — defined by the World Organisation for Animal Health as “the state of the animal…how an animal is coping with the conditions in which it lives” (1). These issues need to be considered by all stakeholders, including veterinarians, to ensure that all parties are aware of the ethical issues at stake and can make a valid contribution to the current debate regarding the creation and use of genetically engineered animals. Read more ...
Building Trust for Engagement of Minorities in Human Subjects Research: Is the Glass Half Full, Half Empty, or the Wrong Size?
Link recommended by Sonia Jiwani

In 1974, in the aftermath of Tuskegee, Congress passed the National Research Act (Pub. L. No. 93-348), creating the National Commission for the Protection of Human Subjects of Biomedical and Behavioral Research as well as US federal regulations providing, for the first time, formal oversight of research with human participants. The Belmont Report, perhaps the most enduring legacy of the Commission, outlined three ethical principles to guide human research, and imposed new requirements for independent institutional review board assessment to ensure that research was consistent with these principles, reasonable with regard to risks and benefits, had appropriate informed consent procedures, and involved vulnerable groups only with justification and appropriate safeguards. Read more ...
Rethinking Research Ethics: The Case of Postmarketing Trials
Alex John London, Jonathan Kimmelman, Benjamin CarlisleLink recommended by Shyam Panthi

From the Nuremberg Code onward, the core mission of human subjects research ethics has been to protect study participants from infringements motivated by a zeal for medical progress. However, with individuals, clinicians, and policymakers increasingly dependent on scientific information for decision-making and with vast social resources invested in developing and utilizing the fruits of research, actors have powerful incentives to coopt research for narrow ends. Contemplated revisions to human subjects research ethics policies in the United States and existing policy in Canada and the United Kingdom fail to capture harms that, although they may not threaten participants, nonetheless undermine the social value of research. This is illustrated by postmarketing (phase IV) research. As a corrective, research ethics should focus on safeguarding the integrity of research as a critical component of an evidence-driven, health information economy. Read more ...
Moral Science: Protecting Participants in Human Subjects Research
Link recommended by Stephanie Cox
he Presidential Commission for the Study of Bioethical Issues today issued its report concerning federally-sponsored research involving human volunteers, concluding that current rules and regulations provide adequate safeguards to mitigate risk. In its report, “Moral Science: Protecting Participants in Human Subjects Research," the Commission also recommended 14 changes to current practices to better protect research subjects, and called on the federal government to improve its tracking of research programs supported with taxpayer dollars. Read more ...
Ethical Considerations in International Research
Link recommended by Stephanie Cox
Physicians, either in their role as investigators or as decision-makers involved in the deliberations related to the funding or the review of research, hold an ethical obligation to ensure the protection of research participants. When the research is to be conducted in countries with differing cultural traditions, health care systems, and ethical standards, and in particular in countries with developing economies and with limited health care resources, US physicians should respect the following guidelines: Read more ...
Boghuma Kabisen Titanji: Ethical riddles in HIV research
TEDXLink recommended by Olga Datskova


FILMED MAY 2012 • POSTED JAN 2013 • TEDxGoodenoughCollege
It’s an all too common story: after participating in an HIV clinical trial, a woman in sub-Saharan Africa is left without the resources to buy a bus ticket to her health clinic, let alone to afford life-saving antiretrovirals. Boghuma Kabisen Titanji asks an important question: how can researchers looking for a cure make sure they’re not taking advantage of those most affected by the pandemic?
Boghuma Kabisen Titanji set out to research HIV drug resistant viruses. In the process, Titanji met a woman who changed the way she thinks about research subjects. Read more ...
Decades Later, Condemnation for a Skid Row Cancer Study
By Gina KolataPublished: October 17, 2013
Link recommended by Nathan Parker


Dr. Perry Hudson, 96, in South Pasadena, Fla. As part of a study he led in Manhattan, biopsies were taken on the prostates of alcoholic men, and testicles were removed in some cases.
A medical researcher from Columbia University, Dr. Perry Hudson, made the skid row alcoholics in Lower Manhattan an offer: If they agreed to surgical biopsies of their prostates, they would get a clean bed and three square meals for a few days, plus free medical care and treatment if they had prostate cancer. Read more ...
Biomedical Research Involving Prisoners
Link recommended by Samantha Rodriguez

Until the early 1970S, R. J. Reynolds, Dow Chemical, the US Army, major pharmaceutical companies, and other sponsors conducted a wide variety of research on prisoners - a captive, vulnerable, and easily accessible population. During that time, approximately 90% of all pharmaceutical research was conducted on prisoners, who also were subjected to biochemical research ranging from testing diet drinks and simple detergents to studies involving dioxin and chemical warfare agents. Read more ...
Henrietta Lacks’ ‘Immortal’ Cells
by Sarah ZielinskiLink recommended by Maleeya Coleman


Henrietta Lacks' cells were essential in developing the polio vaccine and were used in scientific landmarks such as cloning, gene mapping and in vitro fertilization. (Courtesy of the Lacks family)
Medical researchers use laboratory-grown human cells to learn the intricacies of how cells work and test theories about the causes and treatment of diseases. The cell lines they need are “immortal”—they can grow indefinitely, be frozen for decades, divided into different batches and shared among scientists. Read more ...
HEALTHBEAT: Ethical questions abound as babies pioneer research into benefits of gene mapping
by Lauren NeergaardLink recommended by Maleeya Coleman


The healthy baby is part of a large research project outside the nation's capital that is decoding the DNA of hundreds of infants. New parents in a few other cities soon can start signing up for smaller studies to explore what's called genome sequencing - fully mapping someone's genes to look for health risks - should become a part of newborn care.
It's full of ethical challenges. Read more ...
Ethical Violations in International Clinical Trials
A set of links recommended by Sharavanan Ganesan
1. Care and consent: the fraught ethics of international clinical trials
2. Government Accuses Team Led by Harvard Professor of Ethics Violations
3. The Guatemala syphilis experiment, human subjects research abuses, and CAM
Flawed Ethics in HPV Vaccine Trials in India
4. Vaccine trial's ethics criticized
5. Committee investigates ethical violations in HPV vaccine trials
The "Monster" Study
A stuttering experiment performed on twenty-two orphan children in Davenport, Iowa in 1939.
Link recommended by Madhumitha Mahadevan
1. A paper by F. Silverman about the 'Monster' study
2. An article in The New York Times about the 'Monster' study
U.S. Government-Sponsored Mind Control and Tulane
Link recommended by Madhumitha Mahadevan
“By the time I left to go home — just like every time from then on — I would recall nothing of my tests or the different doctors. I would only remember whatever explanations Dr. Robert G. Heath (of Tulane Medical School) gave me for the odd bruises, needle marks, burns on my head and fingers and even the genital soreness. I had no reason to believe otherwise. Already they had begun to control my mind!”
Claudia S. Mullen, March 15, 1995
Testimony to the Advisory Committee on Human Radiation Experiments
Government tested AIDS drugs on foster kids
by John SolomonLink recommended by David Fullerton

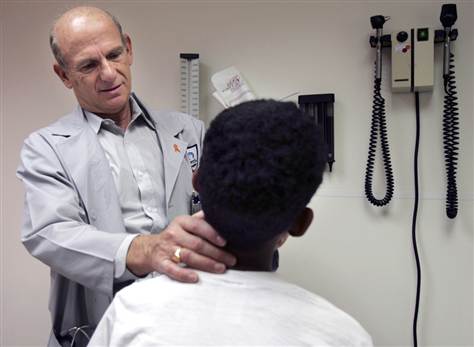
Dr. Ram Yogev, director of the pediatric HIV/AIDS program at Children's Memorial Hospital in Chicago, examines a former foster child who is now adopted, on April 8 in Chicago.
Government-funded researchers tested AIDS drugs on hundreds of foster children over the past two decades, often without providing them a basic protection afforded in federal law and required by some states, an Associated Press review has found.
The research funded by the National Institutes of Health spanned the country. It was most widespread in the 1990s as foster care agencies sought treatments for their HIV-infected children that weren’t yet available in the marketplace. Read more ...
Protecting Human Research Participants
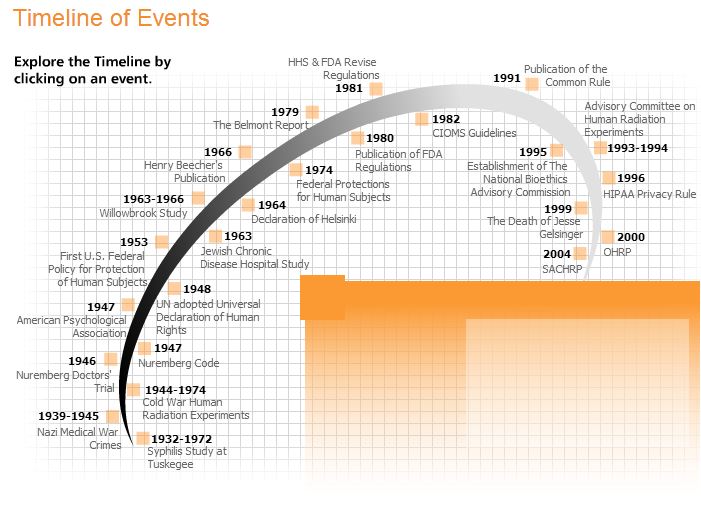
Click the link to visit the interactive website.
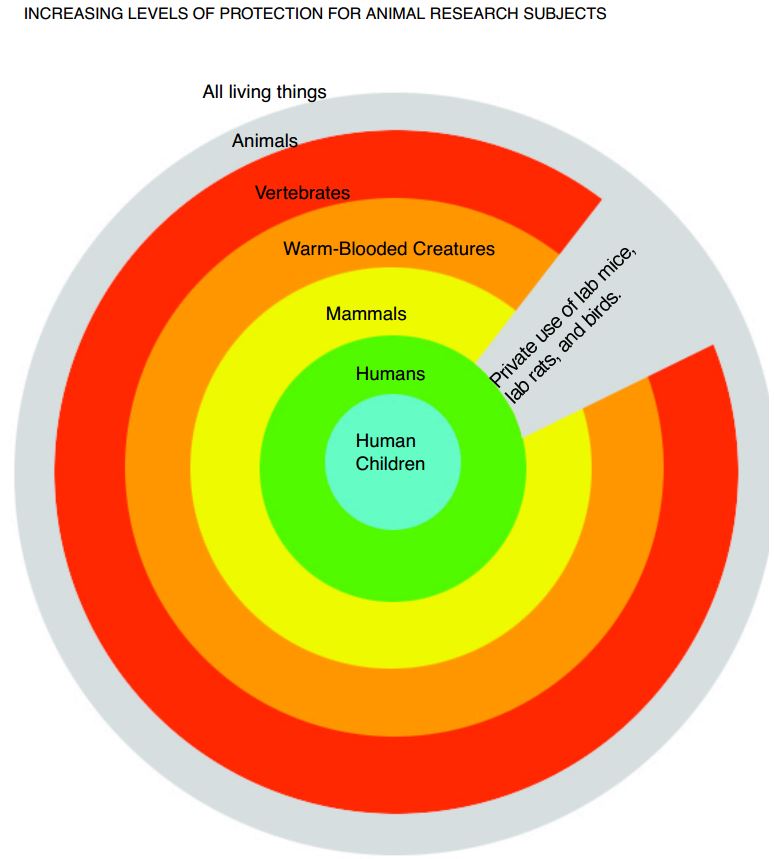
Increasing Levels of Protection For Animal Research Subjects
U.S. Proposes Wildlife Protection for Captive Chimps

Chimp Haven in Keithville, La., serves as the national chimpanzee sanctuary.
By JAMES GORMAN
Published: June 11, 2013
Adding the animals to the list of endangered species would pose a new obstacle to their use in invasive biomedical research. Read more ...
Animal Malpractice: Harvard Shuts Down Primate Center
Link recommended by Malcolm Dcosta

The Harvard primate research center is shutting down after a very troublesome year. Photo credit: nytimes.com
The Harvard primate research center where four monkeys died because of animal care problems will be largely shut down by 2015, Harvard Medical School announced Tuesday afternoon. The university cited a tough economic climate for biomedical research funding and shifting long-term strategic plans.
The announcement stunned outside researchers, because it comes a year and a half after Harvard began investing significant time and resources into the New England Primate Research Center in Southborough, to correct animal care and oversight problems that had resulted in the deaths of four monkeys between June 2010 and Feb. 2012.
The US Department of Agriculture investigated the center and cited Harvard for violations of animal welfare rules, an embarrassing black eye that caused consternation across the university. But medical school leaders said the decision to close the primate center was unrelated to its previous problems. Read more ...
EU bans sale of all animal-tested cosmetics
Link recommended by Malcolm Dcosta

11 March 2013 | The search for alternatives to animal testing goes on
The ban applies to all new cosmetics and their ingredients sold in the EU, regardless of where in the world testing on animals was carried out.
The 27 EU countries have had a ban on such tests in place since 2009. But the EU Commission is now asking the EU's trading partners to do the same.
Animal rights lobbyists said EU officials had "listened to the people".
The anti-vivisection group BUAV and the European Coalition to End Animal Experiments (ECEAE) said they had spent more than 20 years campaigning on the issue and had enlisted celebrities including Sir Paul McCartney, Morrissey and Sienna Miller to their cause. They congratulated the EU Commission for putting the ban into effect. Read more ...
New guidelines announced for risky research

21 Feb 2013 | 19:13 GMT | Posted by Brendan Maher
US government officials have passed two more checkpoints on the long, winding road towards a policy for dealing with risky research. That journey was forced into overdrive at the end of 2011, when a government body recommended against publishing two studies showing how a deadly form of avian influenza H5N1 could be made to pass between mammals.
Today, the US National Institutes of Health (NIH) announced a final framework for vetting specific types of experiments before funding them. The US Office of Science and Technology Policy (OSTP) also published a long-awaited draft policy for how scientists and institutions should monitor and report on a wide range of research that malevolent forces could manipulate to do harm. This type of research, called dual-use research of concern (DURC), is fundable if the potential benefits are deemed significant and the risks deemed manageable. Read more ...
Aligning Regulations and Ethics in Human Research
Rebecca DresserLink recommended by Hawley Kunz

Government officials are revising the 1991 Common Rule regulations that govern most human research in the United States. They have already received public comments on a 2011 Advance Notice of Proposed Rulemaking (ANPRM) (1). The public will have another chance to comment when officials publish specific proposals. The revision effort's overall goal is to remove unwarranted regulatory impediments to research while strengthening essential human subject protections. I offer three ideas for additions to the oversight system, each tied to one of the 1979 Belmont Report's (2) three ethical principles governing human research.
Although prepared by a U.S. advisory group, the Belmont Report expresses values embodied in historic documents like the Nuremberg Code, international statements like the Helsinki Declaration, and domestic oversight systems in countries around the world. The report emphasizes three ethical concepts (see the table). Although these concepts underlie many Common Rule provisions, insights gained since 1991 and unaddressed problems in the current oversight system point to new measures that could enhance the rule's ethical legitimacy. Read more ...
Families Emerge as Silent Victims Of Tuskegee Syphilis Experiment
By Carol Kaesuk YoonPublished: May 12, 1997
Link recommended by Bismah Motawala

It has been 25 years since the nation learned that more than 400 black men infected with syphilis went untreated for decades in a federally financed experiment in this rural Southern town laced with sandy roads and pine forests.
These men, who are expected to receive a Presidential apology on Friday in Washington, have been the subject of countless academic studies, news articles and books, as well as a play and a made-for-television movie. Read more ...
Restricted use of chimps in government research recommended

An Institute of Medicine committee has released a report recommending stringent limits on the use of chimpanzees in biomedical and behavioral lab work performed at the National Institutes of Health. The report comes amid Congressional pressure to make sure such research is completed humanely.
The IOM’s recommendations focus on the scientific need for chimpanzees as research subjects, but they also took ethical issues into consideration. According to the IOM, chimpanzees' genetic closeness to humans and their similar biological and behavioral characteristics not only make these mammals a valuable species for certain types of research but these animals also demand “greater justification for conducting research with them,” the committee said. Read more ...
Studies show 'dark chapter' of medical research
By Elizabeth Landau, CNNOctober 1, 2010 6:08 p.m. EDT
 The Public Health Service took photographs during the Tuskegee syphilis study, but no captions remain. This is one of them.
The Public Health Service took photographs during the Tuskegee syphilis study, but no captions remain. This is one of them.
(CNN) -- The Tuskegee syphilis experiment of the 20th century is often cited as the most famous example of unethical medical research. Now, evidence has emerged that it overlapped with a shorter study, also sponsored by U.S. government health agencies, in which human subjects were unknowingly being harmed by participating in an experiment.
Research from Wellesley College professor Susan Reverby has uncovered evidence of an experiment in Guatemala that infected people with sexually transmitted diseases in an effort to explore treatments. Read more ...
NASA engineer quits over animal experiment

by Hayley Kappes / The Daily News
khou.com
Posted on September 12, 2010 at 1:34 PM
Updated Sunday, Sep 12 at 1:37 PM
JOHNSON SPACE CENTER — April Evans wrestled for weeks whether to quit her dream job working for NASA’s operations with the International Space Station.
She concluded in March there was no other option but to resign after learning the space agency proposed a $1.75 million experiment to irradiate 18 squirrel monkeys to determine astronauts’ ability to withstand high levels of continuous radiation in deep space. Read more ...
Prominent doctors used elderly patients for medical experimentation
Wednesday, October 11, 2006 by: Jerome Douglas
(NaturalNews) Four prominent doctors at Kaplan Hospital in Rehovot and the Hartzfeld Geriatric Hospital in Gedera -- both located in Israel -- were arrested this week on suspicion of using elderly patients as the subjects of hundreds of illegal experiments.
The four doctors are suspected of wrongful death through negligence, abuse of helpless victims, aggravated assault, fraud, violation of a statutory obligation and interference in an investigation. Read more ...
Human medical experimentation in the United States: The shocking true history of modern medicine and psychiatry (1965-2005)
Monday, March 06, 2006 by: Dani Veracity
(1966)
The CIA continues a limited number of MKULTRA plans by beginning Project MKSEARCH to develop and test ways of using biological, chemical and radioactive materials in intelligence operations, and also to develop and test drugs that are able to produce predictable changes in human behavior and physiology (Goliszek).
Dr. Henry Beecher writes, "The well-being, the health, even the actual or potential life of all human beings, born or unborn, depend upon the continuing experimentation in man. Proceed it must; proceed it will. 'The proper study of mankind is man,'" in his "exposé" on human medical experimentation Research and the Individual ("Human Experimentation: Before the Nazi Era and After"). Read more ...











