Authorship and Peer Review Links
Why Trust Science

Do doctors really know what they are talking about when they tell us vaccines are safe? Should we take climate experts at their word when they warn us about the perils of global warming? Why should we trust science when our own politicians don’t? In this landmark book, Naomi Oreskes offers a bold and compelling defense of science, revealing why the social character of scientific knowledge is its greatest strength—and the greatest reason we can trust it. Read more...
Industry Dominates Trump’s New Council of Science Advisers

President Trump revived the President’s Council of Advisors on Science and Technology on Tuesday after nearly two years without it. “Under this administration, science and technology in America continues to advance by leaps and bounds,” said the president’s science adviser, Kelvin Droegemeier, who will chair the council, in a statement. Read more...
Why People Are Marching for Science: ‘There Is No Planet B’

Thousands of people gathered in the rain Saturday on the soggy grounds of the Washington Monument to turn Earth Day into an homage to science. After four hours of speeches and musical performances, they marched down Constitution Avenue to the foot of Capitol Hill, chanting “Build labs, not walls!” Read more...
More South Korean Academics Caught Naming Kids as Co-Authors
Link recommended by Anna An Bui

The number of South Korean academics accused of naming children as co-authors on research manuscripts — even though the children did not contribute to the research — continues to grow. An education ministry report details 11 university academics who named high-school or middle-school-aged children on papers that the children allegedly did not contribute to. Nine of these are newly identified, bringing the total number accused to 17, and the total number of papers affected to 24, since the practice was first exposed in late 2017. Read more...
More Evidence That Nutrition Studies Don’t Always Add Up
Link recommended by Mariajose J Perez

Not too long ago, Brian Wansink was one of the most respected food researchers in America. He founded the Food and Brand Lab at Cornell University, where he won attention for studies that showed that small behavioral changes could influence eating patterns. He found that large plates lead people to eat more food because they make portions look smaller and that children eat more vegetables when they have colorful names like “power peas.” Dr. Wansink wrote best-selling books and published hundreds of studies. For over a year, he served in a top nutrition policy role at the Department of Agriculture under George W. Bush, where he helped shape the government’s influential Dietary Guidelines. His research even led the government to spend almost $20 million redesigning school cafeterias, an initiative known as the Smarter Lunchrooms Movement. Read more...
Research integrity: Don't let transparency damage science
Link recommended by Eugene A Osae

Stephan Lewandowsky and Dorothy Bishop explain how the research community should protect its members from harassment, while encouraging the openness that has become essential to science Transparency has hit the headlines. In the wake of evidence that many research findings are not reproducible, the scientific community has launched initiatives to increase data sharing, transparency and open critique. As with any new development, there are unintended consequences. Many measures that can improve science — shared data, post-publication peer review and public engagement on social media — can be turned against scientists. Read more...
Has a software bug really called decades of brain imaging research into question?
Link recommended by Haley Nicole Smith
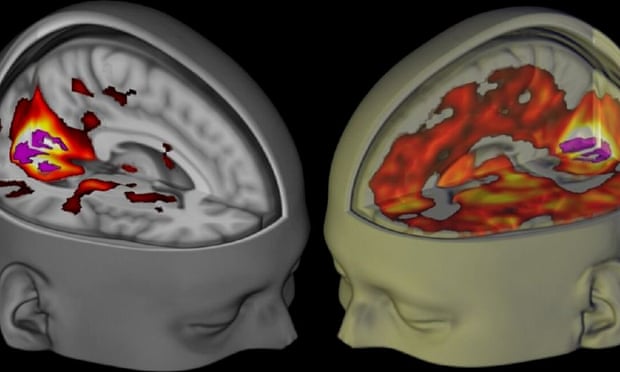
Over the summer, some headlines suggested that a study highlighting issues in the way we analyse fMRI data renders the technique irretrievably flawed. But the reality is much more nuanced. Since its inception in 1990, functional Magnetic Resonance Imaging (fMRI) has revolutionised the way we think about the brain. In association with other techniques, it provides invaluable clues to understand brain disorders, like psychosis or dementia. At its heart, fMRI is a medical imaging technique that allows scientists to look at where oxygenated blood is being directed around the brain – if a brain area is being used for a particular task, it will need more oxygen. So by extension, fMRI allows us to look at how information is processed in the brain, and is one of the few techniques we have to look directly into a person’s brain while they are thinking. Read more...
The Ethical Data Scientist
In the waning months of the Bloomberg administration, I worked for a time in a New York City Hall data group within the Health and Human Services division. One day, we were given a huge dataset on homeless families, which included various characteristics such as the number and age of children and parents, previous ZIP code, the number and lengths of previous stays in homeless services, and race. The data went back 30 years. The goal of the project was to pair homeless families with the most appropriate services, and the first step was to build an algorithm that would predict how long a family would stay in the system given the characteristics we knew when they entered. Read more...
Big Pharma’s Role in Clinical Trials
Link recommended by Sonia Shirwadkar
Pharmaceutical companies are the top sponsors of clinical trials, the best process for determining the safety and efficacy of drugs. Big Pharma funded clinical trials frequently result in publication bias, buried results and misleading information. The pharmaceutical industry is the biggest funder of clinical trials in the U.S., but there is little financial incentive for Big Pharma to be honest about trial results. In the multi-billion dollar industry, companies face pressures from shareholders looking for returns on investments, competitors racing to be the first to develop new treatments, and astronomical costs for researching and developing products. Read more...
Scientific method: Statistical errors
Link recommended by Sahar Anjum
For a brief moment in 2010, Matt Motyl was on the brink of scientific glory: he had discovered that extremists quite literally see the world in black and white. The results were “plain as day”, recalls Motyl, a psychology PhD student at the University of Virginia in Charlottesville. Data from a study of nearly 2,000 people seemed to show that political moderates saw shades of grey more accurately than did either left-wing or right-wing extremists. “The hypothesis was sexy,” he says, “and the data provided clear support.” The P value, a common index for the strength of evidence, was 0.01 — usually interpreted as 'very significant'. Publication in a high-impact journal seemed within Motyl's grasp. But then reality intervened. Sensitive to controversies over reproducibility, Motyl and his adviser, Brian Nosek, decided to replicate the study. With extra data, the P value came out as 0.59 — not even close to the conventional level of significance, 0.05. The effect had disappeared, and with it, Motyl's dreams of youthful fame. Read more...
Fraud Case Seen as a Red Flag for Psychology Research
Link recommended by Sahar Anjum
A well-known psychologist in the Netherlands whose work has been published widely in professional journals falsified data and made up entire experiments, an investigating committee has found. Experts say the case exposes deep flaws in the way science is done in a field, psychology, that has only recently earned a fragile respectability. The psychologist, Diederik Stapel, of Tilburg University, committed academic fraud in “several dozen” published papers, many accepted in respected journals and reported in the news media, according to a report released on Monday by the three Dutch institutions where he has worked: the University of Groningen, the University of Amsterdam, and Tilburg. The journal Science, which published one of Dr. Stapel’s papers in April, posted an “editorial expression of concern” about the research online on Tuesday. Read more...
Good data need good writing
Link recommended by Amanveer Wesley
What place has style in scientific writing? It all depends, one might argue, on where the scientific writing is found. Yet we have all read badly written published papers that are invariably less successful in communicating their ideas. Style is the art of clarity, concision and uniformity of presentation of word or idea, in written form or in picture. The aim of 'good' style is ease of interpretation—to reduce to a minimum the mechanics of the transfer of ideas. As a service to the readers of Nature Immunology, the editors want to take this opportunity to discuss our stylistic requirements. This exercise will emphasize several examples of problems we often encounter in manuscripts that contribute to lack of precision and clarity. Some of these common problems are obvious; some are less so. All, however, cause papers to be less persuasive and impede successful communication of otherwise good data. Read more...
Research misconduct: the poisoning of the well
Link recommended by Sudan Puri
On Wednesday 11 January 2006 Seoul National University concluded that Hwang Woo-suk, a pioneer in stem cell research and a national hero in Korea, had fabricated much of his research. His claim in 2005 to have produced stem cells from adult cells had reverberated around the world because it opened up new ways to treat Parkinson's disease and other degenerative diseases. His disgrace was equally high profile, providing one of the most dramatic cases ever of scientific fraud. Read more...
Report clears researcher who broke drug company agreement
Link recommended by Hanieh Mirhajiianmoghadam
A new report has urged Canada's federal government to do more to curb attempts by pharmaceutical companies to influence the conduct and publication of clinical studies carried out by researchers into their products. The report, commissioned by the Canadian Association of University Teachers, follows a four year dispute at the Toronto Hospital for Sick Children, centring on attempts by a drug company to prevent publication of results of a trial that were unfavourable in relation to one of its products. Read more...
Statisticians issue warning over misuse of P values
Policy statement aims to halt missteps in the quest for certainty.Misuse of the P value — a common test for judging the strength of scientific evidence — is contributing to the number of research findings that cannot be reproduced, the American Statistical Association (ASA) warns in a statement released today. The group has taken the unusual step of issuing principles to guide use of the P value, which it says cannot determine whether a hypothesis is true or whether results are important.
This is the first time that the 177-year-old ASA has made explicit recommendations on such a foundational matter in statistics, says executive director Ron Wasserstein. The society’s members had become increasingly concerned that the P value was being misapplied in ways that cast doubt on statistics generally, he adds. Read more...
Reproducibility will not cure what ails science
Link recommended by Jeeva Babu
Leaders of the scientific community, nudged by the media (including Nature), are acknowledging that a culture of science focused on rewarding eye-catching and positive findings may have resulted in major bodies of knowledge that cannot be reproduced.
Private-sector, academic and non-profit groups are leading multiple efforts to replicate selected published findings, and so far the results do not make happy reading. Several high-profile endeavours have been unable to reproduce the large majority of peer-reviewed studies that they examined. Meanwhile, the US National Academies is preparing to publish a high-profile report on scientific integrity that will flag irreproducibility as a key concern for the research enterprise. Read more...
The Cancer Test
By Jocelyn kaiserLink recommended by Brian Rohde

The email that arrived in Richard Young’s inbox in October 2013 was polite but firm. The writer was part of a group of researchers who “are conducting a study to investigate the reproducibility of recent research findings in cancer biology.” A paper that Young, a biologist at the Massachusetts Institute of Technology in Cambridge, had published in Cell in 2012 on how a protein called c-Myc spurs tumor growth was among 50 highimpact papers chosen for scrutiny by the Reproducibility Project: Cancer Biology. The group might need help with materials and advice on experimental design, the message said. It also promised that the project would “share our procedure” to ensure “a fair replication.” Read more...
Salami publication: definitions and examples
By Vesna Šupak SmolčićLink recommended by Gareth Hastings

Salami publication or segmented publication is a distinct form of redundant publication which is usually characterized by similarity of hypothesis, methodology or results but not text similarity. These aspects of publications are not objectively detected by software applications and therefore present a serious threat to publication ethics. This article presents a practical approach for dealing with manuscripts suspected of salami publication during the submission process and after article publication in Biochemia Medica. Read more...
How Reliable Are Psychology Studies?
By Ed YongLink recommended by Suman Adhikari

No one is entirely clear on how Brian Nosek pulled it off, including Nosek himself. Over the last three years, the psychologist from the University of Virginia persuaded some 270 of his peers to channel their free time into repeating 100 published psychological experiments to see if they could get the same results a second time around. There would be no glory, no empirical eurekas, no breaking of fresh ground. Instead, this initiative—the Reproducibility Project—would be the first big systematic attempt to answer questions that have been vexing psychologists for years, if not decades. What proportion of results in their field are reliable? Read more...
High-impact-factor Syndrome
By Carlton M. Caves
You are surprised to find that you have been tasked with evaluating minor-league pitchers eager to get into major-league baseball. You interview applicants, collect information, and observe their performance. But, being a physicist, you know next to nothing about evaluating pitching skill, so to make your life easier, you fix on a single figure of merit, the pitcher’s heat (fastball speed). Although you have access to each applicant’s fastball speed, you elect to rank the candidates in terms of the average speed of all the pitchers on an applicant’s current minor-league team. Using this as a proxy for individual pitching ability, you assemble a pitching staff. As the season wears on, your pitchers are drubbed in game after game. You see the general manager approaching with a frown on his face, and...the alarm goes off. Read more...
Publishing: The peer-review scam
Link recommended by Zoe Lipowski

Most journal editors know how much effort it takes to persuade busy researchers to review a paper. That is why the editor of The Journal of Enzyme Inhibition and Medicinal Chemistry was puzzled by the reviews for manuscripts by one author — Hyung-In Moon, a medicinal-plant researcher then at Dongguk University in Gyeongju, South Korea.
The reviews themselves were not remarkable: mostly favourable, with some suggestions about how to improve the papers. What was unusual was how quickly they were completed — often within 24 hours. The turnaround was a little too fast, and Claudiu Supuran, the journal's editor-in-chief, started to become suspicious. Read more ...
Asia-Pacific Analysis: Addressing science fraud in Asia
Link recommended by Sarah Jarudi
Is science fraud on the rise in the Asia-Pacific region and what can be done about the problem?
I refer mainly to scientists faking research results, publishing them as factual in science journals and manufacturing endorsements for themselves and their papers. Read more ...
ISU researcher implicated in AIDS research fraud
By Gavin AronsenLink recommended by Sudad Kazzaz

An assistant professor at Iowa State University has resigned after researchers said he falsified research on an AIDS vaccine to make it appear that it could neutralize strains of the HIV virus.
The falsified research came to light after the U.S. Department of Health and Human Services posted a notice Monday revealing the results of a joint investigation by the Office of Research Integrity and ISU that determined Dr. Dong-Pyou Han, an assistant professor in biomedical sciences, contaminated the rabbit blood used in his research with human antibodies. Read more...
Science Journal Pulls 60 Papers in Peer-Review Fraud
Link recommended by Sudad Kazzaz
A scientific journal has retracted 60 papers linked to a researcher in Taiwan, accusing him of “perverting the peer-review process” by creating fraudulent online accounts to judge the papers favorably and help get them published.
age Publications, publisher of The Journal of Vibration and Control, in which the papers appeared over the last four years, said the researcher, Chen-Yuan Chen, had established a “peer-review and citation ring” consisting of fake scientists as well as real ones whose identities he had assumed. It said that in at least one case, Mr. Chen, who also uses the first name Peter, reviewed his own paper using one of the aliases. Read more...
False positives: fraud and misconduct are threatening scientific research
Link recommended by Sophia Ewens

Dirk Smeesters had spent several years of his career as a social psychologist at Erasmus University in Rotterdam studying how consumers behaved in different situations. Did colour have an effect on what they bought? How did death-related stories in the media affect how people picked products? And was it better to use supermodels in cosmetics adverts than average-looking women?
The questions are certainly intriguing, but unfortunately for anyone wanting truthful answers, some of Smeesters' work turned out to be fraudulent. Read more ...
To Get More Out of Science, Show the Rejected Research
Link recommended by Alishpa Masood

In 2013, the federal government spent over $30 billion to support basic scientific research. These funds help create knowledge and stimulate greater productivity and commercial activity, but could we get an even better return on our investment?
The problem is that the research conducted using federal funds is driven — and distorted — by the academic publishing model. The intense competition for space in top journals creates strong pressures for novel, statistically significant effects. As a result, studies that do not turn out as planned or find no evidence of effects claimed in previous research often go unpublished, even though their findings can be important and informative. Read more ...
The scientists who get credit for peer review
By Richard Van NoordenLink recommended by Alishpa Masood


Scientists rarely get credit for one of their most important jobs — the unpaid task of peer reviewing papers. The startup Publons was created to change that by encouraging researchers to post their peer-review history online. Funded with about $US300,000 from angel investors in New Zealand, the company also rewards top reviewers with prizes, such as credits for Amazon Web Services.
On 8 October, Yogendra Kumar Mishra, who studies nanomaterials at the University of Kiel in Germany, was crowned as Publons’ top reviewer for the past three months, on the basis of the volume of reviews he recorded on the site, plus merit points earned for achievements such as reviews being endorsed by others. Read more ...
How to Make More Published Research True
By John P. A. IoannidisLink recommended by Alishpa Masood

The achievements of scientific research are amazing. Science has grown from the occupation of a few dilettanti into a vibrant global industry with more than 15,000,000 people authoring more than 25,000,000 scientific papers in 1996–2011 alone . However, true and readily applicable major discoveries are far fewer. Many new proposed associations and/or effects are false or grossly exaggerated and translation of knowledge into useful applications is often slow and potentially inefficient. Read more ...
Respected medical journal turns to dark side
By Tom SpearsLink recommended by Stefan Madansingh


A respected Canadian medical journal that was sold to offshore owners last year is now printing scientific junk for hire, but still trading on its original good name.
Experimental & Clinical Cardiology was published in Oakville, Ont., for 17 years and had a solid reputation for printing original medical research. It was sold in 2013, and its new owners say they are in Switzerland, but do their banking in Turks and Caicos.
And for $1,200 U.S. they’ll print anything — even a garbled blend of fake cardiology, Latin grammar and missing graphs submitted by the Citizen. Read more ...
Case Study: Should you listen to a peer reviewer?
Link recommended by Stefan Madansingh
ORI will soon release a series of RCR case studies edited by Dr. James Dubois of St. Louis Univerity. The creation of the case studies was funded through ORI's RCR Resource Development program and involved a team of nearly 20 writers, contributors, and reviewers. These well-crafted case studies, along with role playing scenarios, will be available for instructors to incorporate into their institutions' RCR training programs. Below is one of many case studies that will be available. Read more ...
The Office of Research Integrity: Case Summaries
Link recommended by Stefan Madansingh
his page contains cases in which administrative actions were imposed due to findings of research misconduct. The list only includes those who CURRENTLY have an imposed administrative actions against them. It does NOT include the names of individuals whose administrative actions periods have expired. Read more ...
Scientists voice fears over ethics of drug trials remaining unpublished
By Sarah BoseleyLink recommended by Lauren Galley

 Scientists say about 250,000 people have taken part in unpublished trials and have therefore been exposed to all the risks involved in research without the benefits to society they were led to believe would happen Photograph: Image Source / Alamy/Alamy
Scientists say about 250,000 people have taken part in unpublished trials and have therefore been exposed to all the risks involved in research without the benefits to society they were led to believe would happen Photograph: Image Source / Alamy/Alamy
Drug companies and other organisations that carry out clinical trials are violating their ethical obligation to the people who take part by failing to publish the results, scientists will argue on Wednesday.
Almost one in three (29%) large clinical trials in the United States remain unpublished five years after they are finished, according to scientists writing in the British Medical Journal. Of those, 78% have no results at all in the public domain. Read more ...
Research ethics: Zero tolerance
A university cracks down on misconduct in China.By David Cyranoski
Link recommended by Wen-Hao Chou

 Yang Wei wants to reform attitudes towards research ethics at Zhejiang University and across the country.
Yang Wei wants to reform attitudes towards research ethics at Zhejiang University and across the country.
Yang Wei has an easy smile and a carefree, even distracted, air — but he takes such a solemn approach to life that his wife sometimes tells him to relax. “I take everything seriously,” he says.
The former materials scientist certainly took it seriously when, two years after he became president of Zhejiang University (ZJU) in Hangzhou, China, he faced a case of scientific misconduct that became a turning point for his presidency. In early October 2008, the editor of the International Journal of Cardiology discovered that figures in a manuscript by He Haibo, a scientist researching traditional Chinese medicine who had been hired by the ZJU only months before, were suspiciously similar to those in an article that He had published elsewhere. Confronted, He quickly owned up, submitting a 12-page confession to Yang on 26 October. Read more ...
Darwin did not cheat Wallace out of his rightful place in history
By John van WyheLink recommended by Olga Datskova

 Every substantive claim in the popular narrative about Alfred Russel Wallace and evolution turns out to be incorrect. Photograph: Hulton-Deutsch Collection/Corbis
Every substantive claim in the popular narrative about Alfred Russel Wallace and evolution turns out to be incorrect. Photograph: Hulton-Deutsch Collection/Corbis
This year is the centenary of the death of Victorian naturalist and co-discoverer of evolution by natural selection, Alfred Russel Wallace. So we have heard a lot about him recently, including the BBC Two series Bill Bailey's Jungle Hero, an episode of BBC Radio 4's In Our Time, two episodes of The Infinite Monkey Cage and scores of articles, talks and exhibitions. Read more ...
Academic and Industrial Conflict

In an unprecedented case, an internationally renowned computer engineer, who recently left the UC Irvine faculty, has been criminally charged for conflicts of interest involving secret payments from a major Japanese telecommunications firm funding his academic research.
1. The article in VoiceofOC
2. The article in Pacific Standards
Texas cancer agency's $11 million grant bypassed review panels
By Eric Berger, Todd Ackerman | November 29, 2012 | Updated: November 30, 2012 1:58pmLink Recommended by Matthew Cross

The state's cancer agency acknowledged Thursday that it approved an $11 million grant in 2010 - then the largest amount of money it had awarded - without conducting a scientific or commercial review.
The disclosure by the Cancer Prevention and Research Institute of Texas comes after a tumultuous summer in which it had to rescind a $20 million grant to a group led by the University of Texas M.D. Anderson Cancer Center because of questions about the integrity of its review process.
The 2010 grant was given to the Dallas-based biotechnology company Peloton Therapeutics for "recruitment, relocation and formation." Peloton aims to discover and develop new cancer drugs. Its grant was one of the first four commercial grants awarded by the agency. Read more ...
Universities and tobacco money
British Medical Journal Editorial on July 7, 2001Link Recommended by Douglass Diak

In December 2000 Nottingham University announced the establishment of an international centre for corporate social responsibility, with initial funding of £3.8m provided by British American Tobacco (BAT). To protest aganst their university's acceptance of money from the tobacco industry, an MBA student refused to accept his “student of the year” award; Richard Smith, editor of the BMJ, resigned from his post as professor of medical journalism; a cancer research team decided to relocate; and a member of the European parliament relinquished her roles at the university. Read more ...
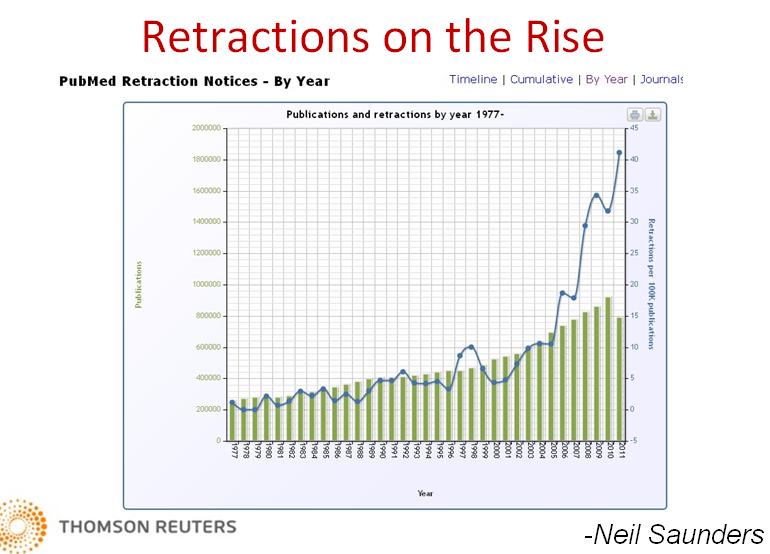
Misconduct, Not Error, Found Behind Most Journal Retractions
By Paul BaskenA set of links recommended by Hawley Kunz


Research misconduct, rather than error, is the leading cause of retractions in scientific journals, with the problem especially pronounced in more prestigious publications, a comprehensive analysis has concluded.
The analysis, described on Monday in PNAS, the Proceedings of the National Academy of Sciences, challenges previous findings that attributed most retractions to mistakes or inadvertent failures in equipment or supplies.
1. The article
2. Commentary by The Chronicle of Higher Education
3. Commentary by the Nature Magazine
A Decade of Misconduct
A senior cardiovascular disease and diabetes researcher at the University of Kentucky has been found guilty of falsifying data over the past 10 years.By Dan Cossins | November 27, 2012

Federal investigators have censured a former University of Kentucky (UK) senior biomedical researcher for serial scientific misconduct over a 10-year period, including the falsification of data in grant applications, progress reports, and published papers. The US Office of Research Integrity (ORI) announced the findings last week (November 20) with a notice in the Federal Register.
A joint investigation carried out over the course of 2 years by the ORI and the UK found that Eric Smart, who studied the molecular mechanisms behind cardiovascular diseases and diabetes, had falsified or fabricated a total of 45 figures—mostly images of Western blots, a technique used to identify proteins—in seven grant applications, three progress reports, and 10 published papers, some of which were cited more than 100 times, according to Thomson Scientific’s Web of Knowledge. The notice says that Smart also reported experimental data from knockout mice that did not exist. Read more ...
The new gatekeepers: reducing research misconduct
 Eugenie Samuel Reich
Eugenie Samuel ReichLink recommended by Erica Soltero

After months of friction that culminated in his openly questioning the reproducibility of data published by his supervisor, a postdoc at the University of Wisconsin–Madison's zoology department was presented with three options. The department's chairman said he could wait to be fired, resign voluntarily or accept a "gracious exit strategy" that would give him time to prepare a paper for publication, if he dropped his "scientific misconduct issues".
When geneticist Aaron Taylor objected that the third option sounded like a "plea bargain" meant to discourage him from pressing his concerns about the lab's data, the chairman, Jeffrey Hardin disagreed. But Hardin also said: "I think you'd have to decide which is more important to you." He later added: "You have to decide whether you want to kind of engage in whistle-blowing. Read more ...
The new gatekeepers: reducing research misconduct
 21 Mar 2012 | 06:26 GMT | Posted by Brendan Maher
21 Mar 2012 | 06:26 GMT | Posted by Brendan Maher
Mistakes, goofs and outright deceptions litter the scientific literature, but there is something that can be done about it. Scientists, writers and journal editors gathered at Rockefeller University in New York last evening to discuss increases in retracted research over the past several years and how best to correct the research record.
“Image manipulation is not a new phenomenon, but it is an increasingly visible one,” said Liz Williams, executive editor of the Journal of Cell Biology (JCB), a Rockefeller University Press journal that has led the way in ferreting out manipulated images before their publication. She was one of three panelists that I helped to bring together for the latest Science Online New York City (SONYC) event, hosted by nature.com and Rockefeller University. Read more ...
A retracted periodontitis-heart disease paper that didn’t make it into the new AHA review

On Wednesday, the American Heart Association announced something that those of us who’d been reading the medical literature carefully had known for a while: Bad gums do not cause heart disease.
Periodontitis is linked to bad heart disease, you see, as studies have shown, and periodontists have sure been using this as an excuse to tell us to floss. But there’s never been a convincing study showing that one causes the other.
In fact, it’s not even clear how you’d do that study. “Let’s see, for a control group, we should have 100 people convince themselves they’re flossing for a year, but not actually floss….oh, what else can we get funding for?” Read more ...
Cracking Open the Scientific Process
An interesting article in The New York Times about new trends in the scientific community.

By THOMAS LIN
Published: January 16, 2012
The New England Journal of Medicine marks its 200th anniversary this year with a timeline celebrating the scientific advances first described in its pages: the stethoscope (1816), the use of ether for anesthesia (1846), and disinfecting hands and instruments before surgery (1867), among others. Read more ...











