
Introduction
History records numerous attempts to determine the age of the Earth. Archbishop Usher of Ireland in 1664 argued that the Earth was "born" on October 26, 4004 BC at 9:00 AM in the morning.
Lord Kelvin (1866) assumed that the Earth was originally molten and that it had taken from 100 million to 20 million years for the Earth to cool to its present temperature distribution. Kelvin's arguments struck a blow at the prevalent geological theories of the time that argued for a much older Earth. Kelvin assumed that there was no source of "new" heat within the Earth.
With the discovery of radioactive isotopes at the end of the previous century Kelvin's hypothesis was abandoned as it was shown that the decay of unstable radioactive isotopes was accompanied by the release of heat energy. Today, most geoscientists believe that the Earth formed about 4.6 billion years ago. It is important to note that the oldest continental crust is approximately 4.0 b.y. and, in general, the volume of rock varies inversely with the age of the rock. That is, with increasing age, the amount of rock of that age decreases. This is supported by the fact that the Earth is a dynamic planet and that the processes introduced in the section on Plate Tectonics have resulted in a recycling of crustal rocks.
Age of the Earth
Hutton (1790) coined the concept of uniformitarianism - "the present is the key to the past". Present processes (such as wind, water, glaciers, etc.) have operated throughout geologic time and that the physical and chemical laws which govern these processes have been operative throughout Earth history. The rates of these processes, however, have not remained constant. For example, land plants first appeared on Earth some 400 million years ago. Think about the effect of moving water and wind across a land surface on which there was no vegetation. Vegetation acts to reduce the effectiveness of these agents of erosion and one would predict that these agents may have been more effective prior to 400 million years ago.
Relative Time
Putting events in their proper order is a common occurrence. I am (probably) older than you and even if we don't tell our actual ages this can be a helpful piece of information. Following Hutton in the late 1700s and into the early 1800s, several important principles were established which made it possible to arrange geologic events into a relative scale.
Application of the Use of the Principles of Relative Age Determination
In a previous chapter it was noted that a great deal of time is most likely missing from a stack of sedimentary rocks. When major (millions of years) amounts of time are missing across a boundary these breaks are called unconformities. From your readings you should be able to define or sketch the following types of unconformities:
- disconformity
- angular unconformity
- nonconformity
An animation to explain the three main types of unconformities - disconformities, non-conformities, and angular unconformities.
When you press the disconformity button, the sequence starts with a period of deposition of horizontal strata. There follows a period of erosion, and then more sedimentation. After choosing non-conformity, the sequence again starts with sedimentation but is followed by uplift and complete erosion of the cover, creating a basement topogrpahy upon which additional strata aare deposited. In the third case, there is a tectonic tilting of early strata before deposition of the later sequence.
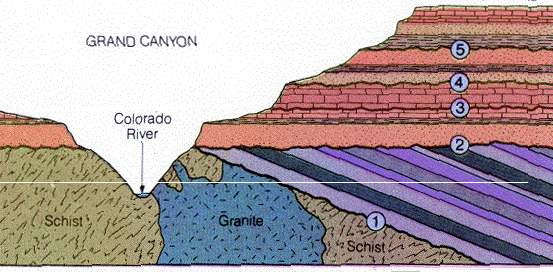
Be able to locate each of these three unconformities on the cross section shown above.
Absolute Time
Radioactive decay is the process whereby an unstable parent atomic nucleus is spontaneously transformed into an atomic nucleus of another element.
- alpha decay - two protons and two neutrons are emitted from the nucleus. This reduces the atomic number of the parent by 2 and the mass number of the parent by 4.
- Beta decay - an electron is emitted from a neutron in the nucleus changing the neutron to a proton. This increases the atomic number of the parent element by 1 but does not change the atomic mass number.
- Electron capture - occurs when a proton captures an electron and changes into a neutron. The atomic number of the parent element is decreased by 1 but the mass number is unchanged.
Heat is also emitted when a parent nucleus undergoes one of these types of decay. Lord Kelvin underestimated the age of the Earth because he was not aware of this internal heat source.
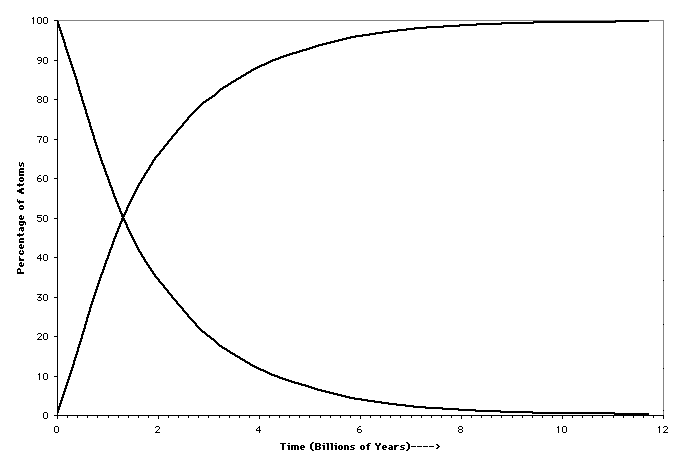
There are several radioactive decay schemes which have been useful in determining the absolute age of geologic events. Each decay scheme can be characterized by the half-life - the length of time it takes for half of the parent atoms to decay to daughter atoms.
For example, if the half life of the decay scheme A goes to B is 100,000 years the changes in amounts of A and B as a function of time are given below, assuming that we start with 1,000,000 atoms of A:
- 1 -100,000 years - 500,000 A and 500,000 B
- 2 -200,000 years - 250,000 A and 750,000 B
- 3 -300,000 years - 125,000 A and 875,000 B
- 4 -400,000 years - 67,500 A and 932,500 B
- 5 -500,000 years - 33,750 A and 966,250 B
- 6 -600,000 years - 16,875 A and 983,125 B
Sketch a plot of time units (number of half-lives) versus the percentage of A atoms remaining. Note that the relationship is not linear. When a decay scheme has gone through 6 or 7 half-lives there is very little of the parent atom left. Elements with relatively short half-lives that were present when the Earth formed have literally decayed themselves out of existence (at least out of the limits of detectability). If these elements decayed to a daughter element that could have been produced by no other decay scheme then we can infer the former existence of the parent from the presence of the daughter.
Animation of the Half-Life Concept explains the concepts of radioactive decay and half life using a
simple cubic array of atoms. The parent element is shown in blue and the daughter in purple. Note how the time for half the atoms to decay remains constant.
Run Eryn Klusko's Simulation of Radioactive Decay and try the "pennies" experiment.
The following parent/daughter pairs have been used to "date" different types of geologic events. In all cases it is necessary to assume that none of the parent was removed in any way other than by radioactive decay. Similarly, one must assume that none of the daughter was removed.
The Geologic Time Scale
Many geologist's work culminated with the development of the Geologic Column. You should "memorize" the column in the text along with several of the important events discussed in lecture. As originally proposed, the Geologic Column allowed assignment of relative ages only. With the discovery of radioactivity, several methods for determining absolute ages evolved. Now, ages can be assigned to the boundaries of the Column.
The figure at the top of this page shows a gabbro sill that has intruded a section of limestone. Relative dating techniques indicate that the sill is younger than the limestone. What we don't know is how much younger the sill is. It could be nearly the same age as the limestone or millions and millions of years younger. If we obtained the absolute age of 100 million years for the gabbro then we would know that the limestone was older than 100 million years.
You should learn the geologic time scale -- at least down to the periods. You should know about when the major mountains of the Earth were finally uplifted and when different organisms appeared and disappeared.