
The Scientific Method
Geology is a body of knowledge strongly tied to experiment and observation. In fact, many observations can be cast as experiments -- thought experiments. Imagine that you are hiking in the Wind River Mountains in Wyoming. At 10,000 feet above sea level you discover a rock containing many shells. Someone notes that some of the shells are remarkably similar to modern clams which live in a shallow marine environment at the present time. Was this area covered by an ocean at some time in the past? Was this ocean 10,000 feet deep? Was this area at one time at sea level and unknown forces elevated it to its present elevation?
The observation has led to the formulation of several hypotheses which attempted to explain the observation. Formulation of a tentative explanation is not sufficient and the investigator will decide what additional information may be needed in order to "prove" the hypothesis. The new information may modify the original explanation or cause it to be discarded. Eventually the investigator may wish to convince others and write a paper or present his or her results at a conference. The process continues and the "explanation" may or may not survive close analysis. A successful hypothesis allows the investigator to make predictions.
Hypotheses become theories when, over time, they meet the challenges presented by newly discovered data and competing hypotheses. A scientific theory must proceed from cause to effect in a logical manner and account for all the relevant facts, not just those that support the theory. It must be consistent with the laws of nature. The revision of scientific hypotheses and theories is considered an integral part of the scientific process.
Using whatever resources you can find, be prepared to answer the following questions:
- What is Occam's Razor?
- How does this compare to the Principle of Parsimony?
Read through this essay on Multiple Working Hypotheses.
During the 17th and 18th centuries the doctrine of catastrophism strongly influenced how people thought about the Earth. According to this doctrine, the Earth's landscape (surface features) was created primarily by great catastrophes -- worldwide floods or fires or some process which we know nothing about because the process no longer operates. The author's note that the doctrine was an attempt to fit the rates of processes to the idea that the Earth was quite young (~ 6,000 years old).
James Hutton, a Scottish farmer and doctor, put forth the principle of uniformitarianism which argues that the physical, chemical and biological laws that operate today have also operated in the geologic past. This doctrine is often stated as follows --- the present is the key to the past. Throughout the course we will have many occasions to return to this principle and use it as a guide for understanding how the Earth works.
In The Beginning
Today the most generally accepted scientific explanation of the origin of the universe is the Big Bang theory. According to this theory, our universe began between 12 and 14 billion years ago with a humongous explosion. Prior to that time all of the matter and energy in the universe were compacted into a single, inconceivably dense point. This is a concept that is difficult to grasp! The universe continues to expand and to cool. During the previous 12 to 14 billion years galaxies, stars and planets have formed from the gas clouds.
The nebular hypothesis argues that diffuse, slowly rotating clouds of hydrogen and helium contract under the force of gravity. Contraction accelerates the rotation of the particles. Matter drifts to the center of the cloud and a proto-Sun may form. Compressed under its own wieght, the proto-Sun becomes hotter and hotter until a temperature is reached where hydrogens fuse to form heliums and energy is released.
Einstein showed that Energy is equal to the square of the speed of light times the mass. A small mass can produce a large amound ot energy.
The following Flash animation shows the collapse of a rotating dust cloud to form a solar system with a central star and orbiting planets. The angular velocity vector is shown in yellow. Escape of light elements to the outer regions is shown immediately after the collapse phase. Show the Animation.
In our solar system, the composition of the planets is a function of distance to the Sun. The inner planets (Mercury, Venus, Earth and Mars) are small and made up rocks and metals. Forming close to the Sun, the low density gasses boiled away and were not retained in quantity.
The outer planets (Jupiter, Saturn, Uranus and Neptune (Pluto is thought to have been captured as a solid object) are sometimes referred to as the gas bags and are made up of ices and gases.
Geological Concepts
For most of you this will be your first close encounter with topics in geology -- or the first since a middle school Earth Science course. However, life experiences have introduced you to geological processes. Earthquakes occur and volcanoes erupt. Basins, like the Gulf of Mexico subside and weathered material is deposited in them by rivers. Sea-level rises and beaches disappear.
Minerals are the materials which make up the solid Earth and rocks are aggregates of minerals. Minerals are made up of atoms of the naturally occurring chemical elements. [These topics are more fully discussed in the following chapters.]
An appreciation for the topics discussed in this course requires a basic familiarity with these Earth materials, i.e. how (and where) they form and what happens to them when exposed at the surface and how may they change if they are buried.
In general, there are three types of rocks:
- Igneous - form by the crystallization of liquid material - magma or lava
- Sedimentary - form in response to conditions at or near the surface of the Earth
- Metamorphic - form in response to changes in temperature and pressure
In the following diagram temperature (increasing to the right) is plotted versus pressure (increasing downwards). The goal is to depict the regions of pressure-temperature space in which the three major rock types occur. There are no hard and fast boundaries. For example, sedimentary processes operate "near the surface of the Earth". With increasing depth of burial, accompanied by an increase in temperature, changes in mineral composition and/or arrangement (texture) begin to modify the material; that is, the sedimentary processes give way to metamorphic processes. The exact temperature and pressure where this occurs depends on too many factors to be able to draw exact boundaries.
At some temperature and pressure the solid material may begin to melt. Rarely does the entire volume of rock melt. A mixture of melt and solids is produced by partial melting. The melt tends to rise (it is usually less dense than the material around it) and is called magma. If the magma is ejected onto the surface it is called lava.
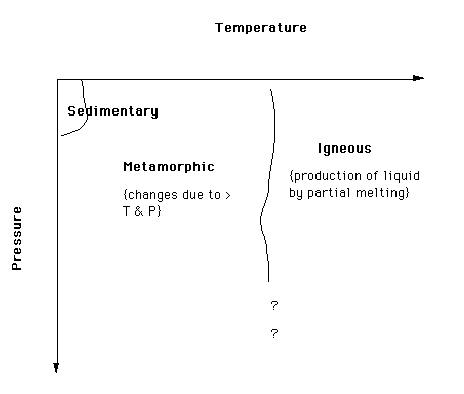
Throughout this course you will encounter several major Earth cycles. An examination of the geologic history of an area might reveal that very old igneous rocks were uplifted, exposed to the Earth's reactive atmosphere, eroded, the debris transported, redeposited and sedimentary rocks were formed. These sedimentary rocks were later buried and metamorphosed. The metamorphic rocks were uplifted and ..... . The cycle does not always follow the same path.
Behavior of Material
A system will be defined as that portion of the universe in which there is an interest. Everything else is part of the surroundings. The system is separated from the surroundings by a boundary.
Physicists have found that they need to specify length, mass and time in order to describe and predict the motions of rigid bodies and fluids. These measures can be described as three fundamental variables.
Every particle of matter in a body (including you and I) is attracted by the Earth and the direction of the force on each particle is toward the center of the Earth. The weight of a body is the gravitational force exerted on it by the Earth. The moon is smaller than the Earth and its gravitational field is less than that of the Earth. You and I would each weigh less on the moon than on the Earth. Our mass, however, is the same on both the moon and the Earth.
Consider two objects of the same size. One is "heavier" than the other. We can compare the two objects by giving their density:
If mass is measured in grams (or pounds) and the volume is measured in cubic centimeters (or cubic feet) then density has the units of g/cc (pounds per cubic foot). If two objects have the same volume (imagine that both are cubes with sides equal to one centimeter), then the one with the greatest mass will have the greater density.
One method for determining the density of an object is to use the Principle of Archimedes. In this method the volume is determined by measuring the apparent loss of weight when a weighed object is immersed in a suitable liquid (we will use water with a density of 1.0 grams/cubic centimeter - g/cc). The object displaces an amount of liquid equal to its own volume and its weight is apparently diminished by the weight of the liquid displaced. If W1 is the weight of the object in air, W2 the weight of the object in water of density 1.0 g/cc, the density is:
Watch the Archimedes Animation and record the weight of the object in air and its weight in water.
Using [1] compute the density of the object.
Do you recall the story of Archimedes? He was asked to determine if a crown was made of pure gold without destroying the crown. Do you see how to apply the information in the animation to his problem?
Often we will need to be able to compare the densities of two objects of different size and shape. The specific gravity of an object is its density divided by the density of an equal volume of water. What are units of specific gravity? Note that the units cancel each other [(g/cc)/(g/cc)] so specific gravity is a dimensionless number.
The density of the continental crust is about 2.7 g/cc. Its specific gravity is 2.7 (since the density of water is about 1.000 g/cc). This means that a unit of continental crust is 2.7 times heavier than an equal unit of water.
One cubic foot of water weighs about 60 pounds. Therefore, one cubic foot of granite (typical continental crust) weighs about 160 pounds [2.7 times 60 pounds]. The specific gravity of gold is about 18. How much does a cubic foot of gold weigh? When I was a kid I saw a movie in which a stagecoach driver picked up a large sack of "gold" and threw it down on the ground. Bah Humbug.
The fourth fundamental variable is the temperature. The temperatures associated with two objects are equal only when the two objects are in thermal equilibrium. Crudely, the higher the temperature of an object the greater the vibration of its constituents atoms. As the temperature is lowered, the degree of vibration decreases. When two bodies at different temperature are brought into contact, heat flows from the warmer to the cooler until the two bodies have identical temperatures - a state of thermal equilibrium is reached.
Note that temperature is not a measure of the amount or quantity of heat. A burning match and a forest fire may have the same temperature but differ markedly in the quantity of heat each possesses. Suppose that two containers of water (a cup and a quart) are placed on identical burners (same temperature) and heated for 10 minutes. The temperature of the cup of water will be higher than the temperature of the quart of water.
Heat can be measured in several units. One calorie is the quantity of heat needed to cause a rise in temperature of 1 o C in one gram of water. Water has a high heat capacity. That is, it takes a lot of heat to raise the temperature of water by a given amount. Similarly, it takes a lot of heat loss to lower the temperature of water. Water, can "store" heat energy more efficiently than rock material.
Imagine a system consisting of a container of water which is hotter at the bottom than it is at the top -- perhaps the system is sitting on a burner on the top of a stove. The system will attempt to even out the variation in temperature so that the system attains thermal equilibrium.
Watch a Convecting Movie and read the first few paragraphs about heat flow.
In general, the density of a material (the ratio of its mass to its volume or M/V) decreases as the temperature increases. Increasing temperature has the effect of increasing the thermal vibration of the atom (or ions or molecules) that comprise the material and it expands (its volume increases). Warm material rises above cooler material of the same composition. Convection may occur when a column of the homogeneous (same composition throughout) material is heated at the bottom. If the material can flow, the warm, lower density material begins to rise to the top of the column, displacing the colder, higher density material. If the column is confined, the colder material sinks, is heated, and begins to rise. Given time, the convection cell works to even out the temperature distribution in the column. Watch a column of water boil. Note that there is a physical movement of material when convection is operative. At elevated temperature and pressure some solids are capable of sustaining this type of flow.
In contrast, heat energy can also be transmitted by conduction in which the temperature contrast is removed by increasing vibration of the atoms (or ions or molecules). Hold a metal knife on a heat source. As most metals are good conductors, it doesn't take long for the knife to become too hot to hold. In general, most rocks are poor conductors.
The Earth is warmed by radiation from the sun. Heat energy is carried by photons emitted from the star. The Earth receives about 10,000 times as much energy from the sun as is generated within the Earth.
We will encounter several situations in which processes are driven by an attempt to redistribute excess heat.
Variations in Composition Within the Earth
The composition of the Earth varies with depth; the Earth is, therefore, not homogeneous but heterogeneous.
- Crust
The crust ranges in thickness from about 2 miles to 40 miles thick. In general, crust underneath the continents is thicker than crust beneath the ocean basins. Continental crust has a density of about 2.6 g/cc as compared to about 2.9 g/cc for the oceanic crust. The different densities reflect different a difference in mineralogy.
- Mantle
The mantle lies beneath the crust and extends to a depth of about 1800 miles. The mantle is denser than the crust (about 3.2 g/cc).
- Core
The core is made up primarily of iron and nickel. The outer portion of the core is liquid and the inner portion is solid. The core has an average density of about 11 g/cc
The average density of the Earth is about 5.5 g/cc.
Variations in Behavior of the Earth
- Lithosphere
The outer most rigid part of the earth includes the crust and upper part of the mantle. Although this interval includes materials of markedly different chemical composition, they act as a unit and respond similarly to mechanical deformation.
The lithosphere averages about 60 miles in thickness but may be more than 150 miles thick beneath the uplifted portions of continents.
- Asthenosphere
The asthenosphere lies beneath the lithosphere and extends to a depth of about 400 miles. A small amount of liquid (derived by the partial melting of the upper mantle) occurs in the upper 100 miles of the asthenosphere; the lithosphere is effectively detached from the underlying asthenosphere. The asthenosphere is weak relative to the strong lithosphere and is capable of flowing - convective.
Plate Tectonics
Plate tectonics was the first comprehensive model of the Earth's internal workings. The history of the hypothesis of Continental Drift is a good place to begin your reading.
Eventually this hypothesis was rejected by most geoscientists and the plate tectonic hypothesis evolved. According to this model the lithosphere is broken into numerous segments called plates which are in motion relative to each other and constantly changing shape and position. There are approximately 12 major plates and many smaller ones. The plates move slowly at a rate of 2 to 12 cm per year driven by the unequal distribution of heat within the Earth. Concepts from plate tectonics will serve as a frame of reference throughout this course.
Plates are bounded by some combination of three boundary types:
- Divergent
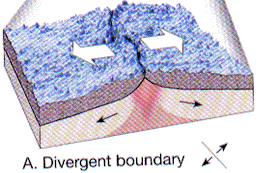 Plates move apart resulting in the upwelling of material from the asthenosphere to form new oceanic crust at spreading centers. As new lithosphere is formed it is slowly displaced away from the ridge axis. It begins to cool and contract and increases in density.
Plates move apart resulting in the upwelling of material from the asthenosphere to form new oceanic crust at spreading centers. As new lithosphere is formed it is slowly displaced away from the ridge axis. It begins to cool and contract and increases in density.
Find a map of the Earth on which water depths are color coded. Note that along spreading centers - such as the Mid-Atlantic Ridge, water depths are low relative to depths away from the spreading center. Iceland sits atop the Mid-Atlantic Ridge.
Bullard discovered that the continents fit together much better if you include their continental
shelves. He limited gaps and overlaps to 4% - easily explicable by vulcanism (e.g. Davis Volcanics in the Labrador Trough) and local crustal deformation. Try this animation to see the fit made using the continental shelves.
This animation was constructed from USGS age and bathymetry maps for the North and South Atlantic Oceans and surrounding continents. The youngest rocks (red colors) clearly outline the mid-ocean ridge system, complete with transform faults. The oldest ocean crust, colored blue is confined to offshore regions adjacent to the United States, Canada, and western Africa. This distribution clearly demonstrates that the North Atlantic began to open before the South Atlantic. Note the goodness of fit of the conjugate continental shelfs as compared with the coastlines.
- Convergent
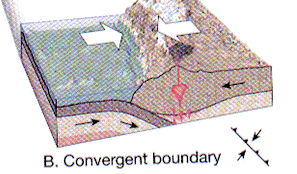 New lithosphere is constantly being added at spreading centers but the Earth's volume remains constant. Therefore, older lithosphere must be being reduced. At convergent margins two plates move toward each other. The interaction is a function of the densities of the two plates:
New lithosphere is constantly being added at spreading centers but the Earth's volume remains constant. Therefore, older lithosphere must be being reduced. At convergent margins two plates move toward each other. The interaction is a function of the densities of the two plates:
- ocean - continental
The oceanic crust bends downward and the continental crust overides it forming a subduction zone and a oceanic trench -- the west coast of South America, for example.
- ocean - ocean
One piece of oceanic crust is subducted beneath the other forming island arcs such the Aleutian Islands
- continent - continent
Continental curst cannot be subducted - its low density causes it to "float". Where two continental plates come together the plates thicken and are sutured together; the Himalayan Mountains, for example.
- Transform Fault
 Two plates move past one another without generating new lithosphere or consuming old lithosphere. The San Andreas fault in California has been studied for many years. Along this fault the Pacific plate (containing San Diego) is moving to the northwest past the North American Plate (containing San Francisco) which is moving to the west. The movement is not even and periodically energy is released (earthquakes occur) when rock are broken at depth.
Two plates move past one another without generating new lithosphere or consuming old lithosphere. The San Andreas fault in California has been studied for many years. Along this fault the Pacific plate (containing San Diego) is moving to the northwest past the North American Plate (containing San Francisco) which is moving to the west. The movement is not even and periodically energy is released (earthquakes occur) when rock are broken at depth.
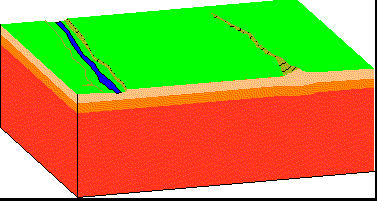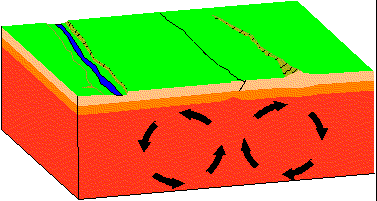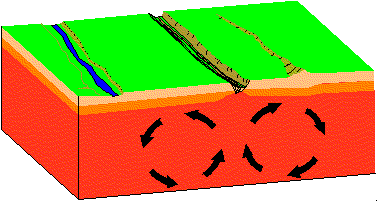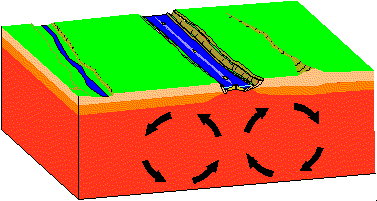 In the animation above a "rift" or "tear" develops where the two convection cells pull apart. With time the rift opens wider and becomes deeper. If there is a marine body of water nearby, the marine waters may enter the rift and a "new ocean" may form. If you hit Reload on your browser you can start the simulation.
In the animation above a "rift" or "tear" develops where the two convection cells pull apart. With time the rift opens wider and becomes deeper. If there is a marine body of water nearby, the marine waters may enter the rift and a "new ocean" may form. If you hit Reload on your browser you can start the simulation.
_________________________________________________________________________________________________
| jbutler@uh.edu
|ClassListserv
|Textbook Home Page
|Glossary of Geologic Terms|
|Search These Pages|Other Courses|Resources|Grade Book|
_________________________________________________________________________________________________
Return to Chapter 1
Copyright by John C. Butler, July 29, 1995
Updated - April, 2000